This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study reveals target for reversing scar tissue after heart attack
A new study by investigators at Massachusetts General Hospital (MGH) reveals an important step to help the human heart regenerate after myocardial infarction (MI).
For the first time, the researchers compared the formation of scar tissue in the injured hearts of zebrafish and mice, discovering how to potentially reverse the permanent scarring that is so damaging to mammal hearts. The results of the study are published in Nature Communications.
"We are the first to directly compare and show very fundamental differences in the formation of scar tissue between zebrafish and mammals," says lead author Eman A. Akam-Baxter, Ph.D., an investigator at the MGH Cardiovascular Research Center and instructor in Medicine at Harvard Medical School. "The results of our study point to a possible new target to reverse scarring after myocardial infarction, which has never been shown before."
A myocardial infarction causes the death of massive numbers of heart cells. To repair the injury, the body replaces the damaged and dead cells with scar tissue. Initially, the scar tissue is beneficial in keeping the heart intact. But eventually the scar tissue becomes a permanent part of the cardiac muscle, causing the heart to pump blood less effectively. An overworked heart causes the scar tissue to expand, leading to permanent heart damage.
The formation of permanent scar tissue after a heart injury is characteristic of all mammals. But zebrafish have a remarkable ability to completely remove scar tissue after an injury, allowing space for cardiac cells to regenerate and fully regrow a healthy heart.
"For many years, investigators have focused on the properties of the cardiomyocytes (heart muscle cells) and immune cells in the zebrafish heart to explain this phenomenon" explained David Sosnovik, MD, the senior author of the paper.
"However, no studies characterizing the nature of the collagen scar in zebrafish have been performed. The expertise of Dr. Akam-Baxter in synthetic and analytical chemistry allowed us to approach this problem from a new angle."
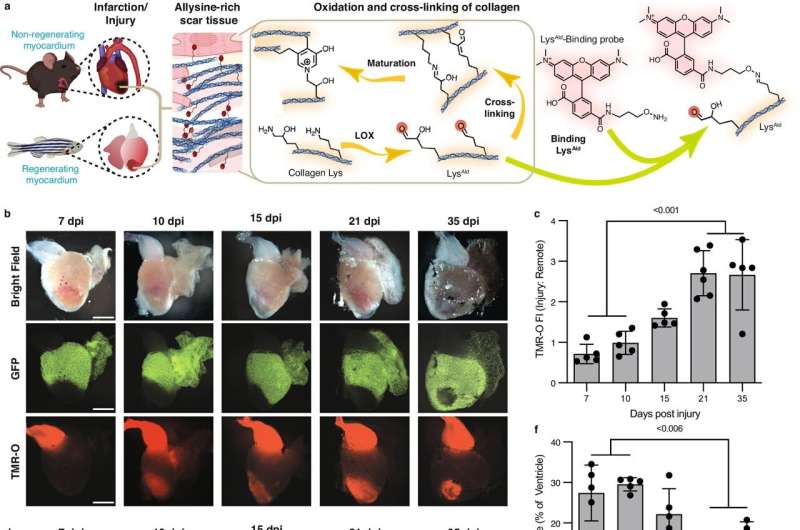
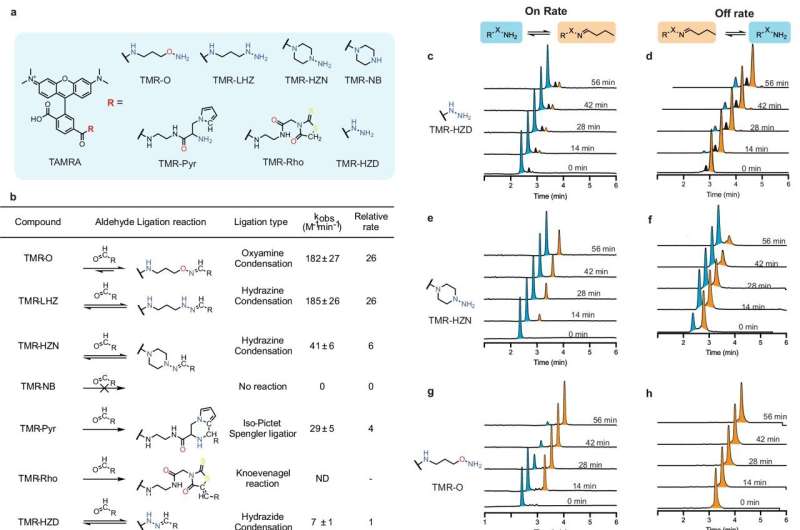
Until now, it was not possible to examine scar tissue formation in the tiny zebrafish heart by imaging. For this study, the researchers first had to develop a molecular imaging probe they named TMR-O, which allowed them to see details of the scarring inside the hearts of zebrafish and mouse models of cardiac injury.
Scar tissue is comprised of collagen, long strands of protein that bind to each other to form a fiber that gives scar tissue its structure and stability. The process of binding of collagen molecules is called cross-linking.
"Think of cross-linked collagen as a network of long strands of protein linked together," explains Akam-Baxter. "Hands on multiple points of each protein strand clasp the opposite strand like a handshake."
The probe the researchers developed bound to each hand, providing a fluorescent readout of how the collagen was cross-linked.
Researchers have long believed that the extent of collagen cross-linking is the key to whether a scar is resorbable or permanent. But when the MGH investigators tested that hypothesis, they found that the amount of cross-linking was similar in the zebrafish and the mice after cardiac injury. However, the type of cross-link was different.
"In the mouse heart, the chemical nature of the collagen cross-links was highly mature, and formed a structure that cannot be broken down by the antifibrotic enzymes in the body. In contrast, the cross-links in the zebrafish resembled a looser handshake," says Akam-Baxter. "The cross-links in the zebrafish heart persisted in a chemically immature form that can be broken down, and this allowed the fibrotic scars to be resorbed and replaced with regenerated heart cells."
The authors further showed that the cross-links that form in the mouse hearts are a result of chemical modification (lysine hydroxylation) of the collagen strands in mice, and that this doesn't occur to the same extent in the zebrafish heart.
This modification is made by an enzyme called lysyl hydroxylase 2; this enzyme is linked to permanent scarring in other organs in diseases of fibrosis.
"No one has studied the effect of blocking this enzyme in the context of heart attack," says Akam-Baxter. Her research team is studying whether inhibition of this enzyme can effectively prevent permanent scarring in the heart after an MI. The researchers will also investigate whether scar tissue can be reversed in other organs.
"The numbers of deaths and amount of heart failure that results from scarring after MI are staggering," says Akam-Baxter. "And fibrotic diseases also account for a huge number of deaths. If we can find a common feature of reversing scar tissue in multiple organs, we can potentially save many lives."
More information: Eman A. Akam-Baxter et al, Dynamics of collagen oxidation and cross linking in regenerating and irreversibly infarcted myocardium, Nature Communications (2024). DOI: 10.1038/s41467-024-48604-7