This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
TCR CDR3s and renalase-1 linked to increased melanoma survival
A new research paper titled "Chemical complementarity of tumor resident, T-cell receptor CDR3s and renalase-1 correlates with increased melanoma survival" has been published in Oncotarget.
As mentioned in the Abstract of this study, overexpression of the secretory protein renalase-1 negatively impacts the survival of melanoma and pancreatic cancer patients, while inhibition of renalase-1 signaling drives tumor rejection by promoting T-cell activation.
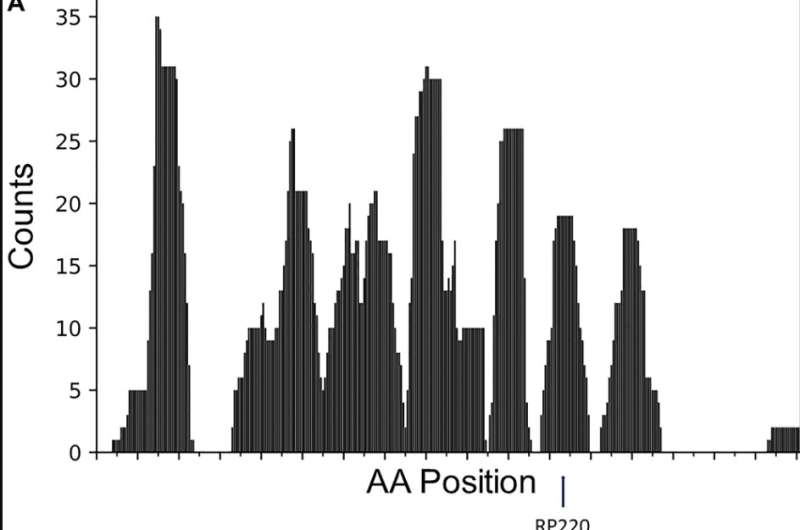
Thus, researchers Saif Zaman, Fred S. Gorelick, Andrea Chrobrutskiy, Boris I. Chobrutskiy, Gary V. Desir, and George Blanck from Yale School of Medicine, Veteran's Administration Healthcare System, Oregon Health and Science University Hospital, Morsani College of Medicine, and H. Lee Moffitt Cancer Center and Research Institute, investigated the chemical complementarity between melanoma-resident, T-cell receptor (TCR) complementarity-determining region 3 (CDR3) amino acid sequences (AAs) and the renalase-1 protein.
"In this study, we asked whether the RNLS protein could potentially be a tumor antigen by examining chemical complementarity between melanoma tumor-resident TCR CDR3s and the AA sequence of RNLS," the researchers write.
The results suggest that there could be biologically relevant antigenic interaction between RNLS epitopes and T-cell receptors (TCRs).
"We hypothesize that RNLS protein could be recognized by TCRs, leading to local immune responses against melanoma, similar to what we have previously demonstrated with wildtype cancer antigens in the melanoma and glioblastoma settings," the researchers explain.
More information: Saif Zaman et al, Chemical complementarity of tumor resident, T-cell receptor CDR3s and renalase-1 correlates with increased melanoma survival, Oncotarget (2024). DOI: 10.18632/oncotarget.28633