November 11, 2011 report
Mystery of resistance to malaria solved in new study
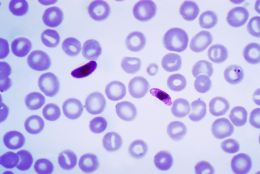
(Medical Xpress) -- Malaria is a disease caused by parasites passed to humans via the bites of infected mosquitoes. Globally, the disease causes over a million deaths every year, and is especially rife in parts of Africa and Asia. The parasites infect red blood corpuscles (the hemoglobin-containing cells that carry oxygen around the body) and hijack the support structure within the cells. Some people are known to be naturally resistant to the serious effects of malaria, and scientists have wondered for decades exactly how their resistance functions. Now new research gone a long way to solving the mystery.
It has been known for decades that some people in Africa and elsewhere who have a mutated gene that causes sickle cell anemia also have resistance to malaria because their red blood corpuscles contain an unusual form of hemoglobin―hemoglobin S, which results in the hemoglobin aggregating within the cell. Possessing only one copy of the mutated hemoglobin S makes the person a largely asymptomatic carrier, while two copies produces symptomatic sickle-cell anemia. In both cases the mutation gives some protection against malaria. Another mutation, hemoglobin C, causes hemolytic anemia when two copies of the mutation are present, and this form also protects against malaria.
In a paper published in Science, researcher Marek Cyrklaff, of Heidelberg University in Germany, and colleagues in Germany and Burkina Faso, report that the unusual forms of hemoglobin in the red cells prevent the malaria parasite, Plasmodium falciparum, from hijacking the actin filaments that provide the skeleton scaffolding within the cell. They compared healthy and infected red corpuscles containing 'normal' hemoglobin with healthy and infected cells containing hemoglobin S or hemoglobin C. Using powerful cryoelectron tomography, the scientists discovered that in normal healthy cells the filaments of actin protein are short and located beneath the outer cell membrane , where they provide a support structure for the cell and makes it strong but pliable enough to pass through the tiniest blood vessels.
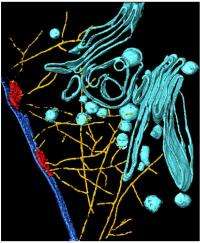
In infected cells with normal hemoglobin they found the actin protein was in long filaments, which the parasite used to build a cytoskeleton, or intracellular bridge, within the cell to transport its own manufactured protein, adhesin, to the surface of the cell. The effect of adhesin, as its name suggests, is to make adjoining cells stick together and to stick the cells to the blood vessel walls, causing the inflammation responses characteristic of malaria. In the hemoglobin S and C cells, the bridge could not be completed and the adhesin could not be effectively transported to the cell surface, thus reducing cell stickiness.
The scientists also found, after further experiments, that hemoglobin C and S are more easily oxidized than the unmutated form, and when actin filaments were placed with the hemoglobin, the C and S forms resulted in shorter actin filaments than normal hemoglobin, as did oxidized hemoglobin.
Malaria is most often treated with quinine, but clinical trials of a vaccine are now being carried out in Africa by GlaxoSmithKline, and the results look promising, with a 65% effectiveness rate. The new research suggests that further drugs could eventually be developed that interfere with the parasite's ability to use the actin filaments for its own purposes.
More information: Hemoglobins S and C Interfere with Actin Remodeling in Plasmodium falciparum–Infected Erythrocytes, Science, DOI: 10.1126/science.1213775
ABSTRACT
The hemoglobins S and C protect carriers from severe Plasmodium falciparum malaria. Here, we found that these hemoglobinopathies affected the trafficking system that directs parasite-encoded proteins to the surface of infected erythrocytes. Cryoelectron tomography revealed that the parasite generated a host-derived actin cytoskeleton within the cytoplasm of wild-type red cells that connected the Maurer's clefts with the host cell membrane and to which transport vesicles were attached. The actin cytoskeleton and the Maurer's clefts were aberrant in erythrocytes containing hemoglobin S or C. Hemoglobin oxidation products, enriched in hemoglobin S and C erythrocytes, inhibited actin polymerization in vitro and may account for the protective role in malaria.
(c) 2011 Medical Xpress

















