Secrets of immune response illuminated in new study
When disease-causing invaders like bacteria infect a human host, cells of various types swing into action, coordinating their activities to address the threat.
In new research appearing in this month's issue of the journal Nature Immunology, Roy Curtiss, director of the Center for Infectious Diseases and Vaccinology at the Biodesign Institute at Arizona State University, along with international collaborators, investigates the coordination of a particular type of immune response, involving the release of of IFN-λ— a cell-signaling protein molecule known as a cytokine.
Molecules like IFN-λ have long been recognized as vital weapons in the immune system's arsenal against viral, bacterial and parasitic pathogens, as well as tumors. They are known as interferons—named for their ability to interfere with the functioning or replication of infectious agents. Communication between cells enabled by interferons can trigger the protective defenses of the immune system, which will attempt by various means to eradicate the infectious pathogen.
"The inception of this study was based on studies conducted in collaboration with the Richard Strugnell group at the University of Melbourne when it was shown that flagella produced by S. Typhimurium—and especially by a mutant generated by Shifeng Wang in our group, that hyper produced the flagellin—were superior in inducing a cascade in host cells leading to the production of NFκB," a protein complex that plays a key role in regulating the immune response to infection.
The cytokine IFN-λ is produced by a type of lymphocyte known as a memory CD8+ T cell. Memory T cells are a vital part of the adaptive immune system. Typically, they are activated and induced to proliferate when they come in contact with a specific antigen produced by the infectious agent and recognized by the T cell's antigen receptor. After their initial encounter with the unfamiliar invader, memory T cells survive in the host in an inactive state, "remembering" the cognate antigen to which they are related. Should they re-encounter this antigen, they can speedily mount a response, liberating IFN-λ.
An understanding of how IFN-λ release is regulated and the complex pathways involved in the production of this key cytokine remains incomplete. The current study demonstrates that the release of IFN-λ by memory T cells can also occur without the activation of these cells by direct contact with the disease antigen. In this way, memory CD8+ T cells also contribute to the host's innate immune response.
The mechanism for this antigen-independent immune response is the focus of the current study. The team's results significantly advance the understanding of such pathways and their subtle regulation, and may stimulate new biomedical approaches to interfering with and disabling disease-causing intruders.
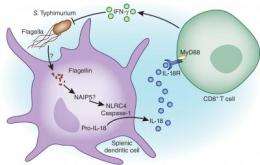
In the new study, the group found that the antigen-independent production of IFN-λ by memory T cells relies on another cell type, known as splenic dendritic cells. Such cells contain so-called NOD-like receptors (NLRs). The NLR's are able to sniff out pathogen-associated molecular patterns. When they sense these distinctive patterns, the NLRs sound the alert.
While their more familiar cousins, the TOLL-like receptors, sense pathogen-associated molecular patterns in the extracellular space, NLRs sense pathogenic traces in the intracellular compartments. Further, once NLR's have successfully detected their target, they assemble large protein complexes in the dendritic cell, known as inflammasomes.
In the case of bacterial invasion, the NLR inside the splenic dendritic cell is triggered when it senses flagellin—a protein associated with bacterial flagellum. The NLR then assembles the inflammasome complex, which produces two key pro-inflammatory interleukins—IL-1 and IL-18. It is the second of these that will migrate from the dendritic cell to the memory CD8+ T cell, triggering the release of IFN-λ. Figure 1 graphically describes this process.
In the current study, antigen-independent secretion of IFN-λ by memory CD8+ T cells was demonstrated in mice infected with the intracellular pathogen Salmonella Typhimurium. The response could be detected as soon as 2 hours post-infection—the NOD-like receptors representing the earliest response to pathogenic invasion. Further, by using strains of S. Typhimurium deficient in flaggelin, the group showed impaired IFN-λ secretion by CD8+ T cells.
Intriguingly, the group also found a robust IFN-λ response when dendritic cells were presented with heat-killed S. Typhimurium or even with the injection of purified flaggelin alone—powerful evidence that the dendritic cell inflammasome assembled by the NOD-like receptor's sensing of flaggelin was sufficient to induce IFN-λ production in memory CD8+ T cells. This flaggelin-induced response was also demonstrated for two other pathogens: Yersinia and Pseudomonas.
The study also confirmed the hypothesis that production of IL-18 in dendritic cells, following inflammasome formation, generated the production of IFN-λ by attaching to a specific receptor-adaptor on the memory CD8+ T cells, which the group identified. The authors speculate that a particular inflammasome known as NAIP5 in splenic dendritic cells is responsible for sensing flaggelin and initiating the cascade of events leading to IFN-λ production in CD8+ T cells.
Previous research had suggested a mechanism for S. Typhimurium to transfer flagellin into the cell from the extracellular medium, through the pathogen's specific secretion system. This is considered critical, as many bacteria are non-pathogenic and indeed, important to the host. An inflammasome response to these so-called commensal bacteria could therefore have disastrous consequences, triggering an inappropriate auto-immune response.
Experiments also demonstrated that not all bacterial flagellins are recognized by the inflammasomes—E. coli flagellins, for example, are not. The reasons for this have yet to be fully explored. The team further speculates that the inflammasome system and its NOD-like receptors may have evolved not only to deal with pathogenic invaders but to carefully regulate the balance of commensal bacteria, keeping populations of healthy microbes in check.
The study was the fruit of a multi-institute collaboration, co-authored by researchers from Department of Microbiology and Immunology, The University of Melbourne, Australia; Department of Biochemistry, University of Lausanne, Epalinges, Switzerland; Department of Infectious Diseases & Pulmonary Medicine, Charité University Hospital, Berlin, Germany and the Ludwig Institute of Cancer Research, Heidelberg, Australia.
As Curtiss explains, the group's research findings lay the groundwork for future investigation: "We are now incorporating the findings from the current study to design a superior recombinant attenuated Salmonella vaccine strain with greatly enhanced ability to induce a CD8-dependent cellular immunity against viral, parasitic and bacterial pathogens in which a CD8 response is critical for successful control."















