Hunting for the last remaining tumour cell
The 7.5 millilitres of blood contained in a standard sample tube is not nearly enough to detect circulating tumour cells (CTCs) in the blood of patients with metastatic breast cancer, prostate cancer, or colorectal cancer with a high enough degree of reliability. These CTCs are the cause of metastases in cancer patients. If they can be effectively characterized, it is possible to arrive at a prognosis and to determine the most effective course of treatment. However, the only way to detect "that one particular cell" is to subject the patient's entire blood volume to a kind of dialysis. This was the conclusion reached by Dr Frank Coumans in an article published in the journal Clinical Cancer Research. Dr Coumans works at the University of Twente's MIRA Institute for Biomedical Technology and Technical Medicine. He was recently awarded a PhD (with distinction) on this topic.
The standard test for circulating tumour cells (CTCs) involves a 7.5 millilitre blood sample and the use of magnetic particles to separate CTCs from the other components of the blood. However, Dr Coumans found that this method failed to detect any CTCs in nearly half of those patients who are known to have metastases. Accordingly, for these patients, this method is an insufficiently reliable indicator of metastases. Furthermore, if no CTCs can be isolated, it will not be possible to develop treatment plans that are tailored to individual patients.
Difficult to identify
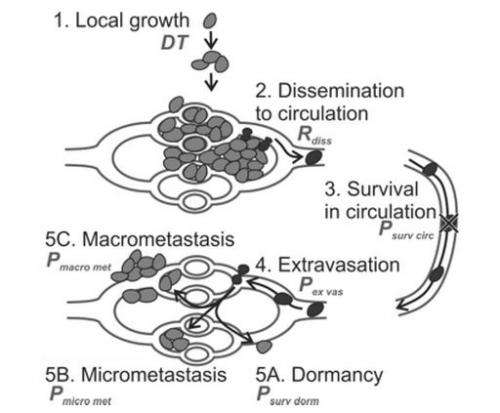
There are several reasons for this. It is no easy matter to pick out CTCs, as they resemble many of the cells that are normally found in the blood. For this reason, the test focuses on specific indicators. One of these is the epithelial adhesion molecule (EpCAM), which "sticks cells together" in organs, but which is not normally found in blood cells. Other such indicators are cytokeratin and CD45, a protein found in all white blood cells. Any white blood cells that unexpectedly contain traces of EpCAM and cytokeratin but very little CD45 could mistakenly be counted as circulating tumour cells. Errors can also creep into the count during the enrichment of CTCs from blood using magnetic particles. For instance, the magnetic field used might be too weak to capture the CTCs. Finally, if the concentration of CTCs is sufficiently low, there is only a small chance that any of these cells will be present in the blood sample. The latter turned out to be the case. Taking two blood samples rather than one has no effect whatsoever. The concentration of CTCs is logarithmic, so doubling the sample size does not double the chances of finding one of these cells.
Large volume
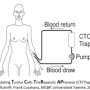
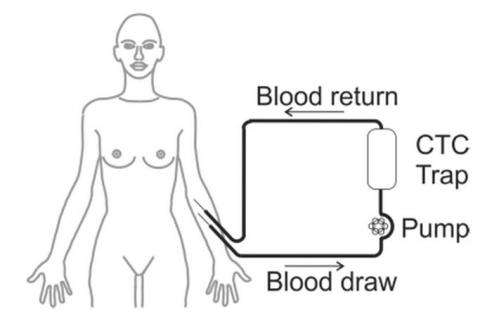
Frank Coumans carried out his PhD research in Prof. Leon Terstappen's Medical Cell Biophysics group. Research previously carried out by this group showed that gains can still be made, for example, by using an improved CellSearch analyser, with better illumination. However, according to Dr Coumans, the only way to achieve a 99 percent chance of detecting a CTC is to sample a blood volume of five litres. This approach would involve the use of additional technology. This works best with fresh blood, so a type of dialysis or apheresis is needed that can separate specific components from the blood and return "the rest" to the patient. This is known as the CTCtrap or CTC TheRapeutic APheresis.
Dr Coumans' research is part of a major European research programme (www.utwente.nl/tnw/ctctrap/). He expects this programme to deliver a technique capable of entering the clinical phase. Metastases often dramatically worsen the prognoses of cancer patients. It is therefore vital to improve the chances of a CTC "hit" and to better understand the mechanisms involved.
The article entitled "Challenges in the Enumeration and Phenotyping of CTC" by Frank Coumans, Sjoerd Ligthart, Jonathan Uhr and Leon Terstappen, was published in Clinical Cancer Research, the journal of the American Association for Cancer Research (ACCR).