Recently discovered stem cell population could one day provide useful source material for kidney repair
Within every human kidney, millions of filtration units known as nephrons are hard at work clearing metabolic waste products from the blood. Given the dirty work they perform, one might expect that the cells composing the nephrons undergo routine self-replacement, but nephrons retain very limited regenerative capabilities and essentially shut down when those limits are exceeded—a potential prelude to organ failure.
"Identification of stem cells in the kidney is of paramount importance if we are to better understand their contribution to kidney disease and harness their regenerative medicine potential," says Nick Barker of the A*STAR Institute of Medical Biology, Singapore. Barker's team has now made important progress towards this goal, identifying cells that appear to be critical progenitors for several structures within the mammalian nephron.
Previous studies have revealed the general cell pool from which these structures emerge, but not the specific cell subsets within that pool that directly contribute to nephron formation. Barker and colleagues were therefore interested in identifying specific proteins that might 'mark' such cells. Since his team had recently identified a gene called Lgr5 as a marker for key stem cell pools in several other major organ systems, they attempted to determine whether this same gene may also be relevant in the early stages of kidney formation.
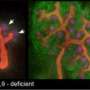
Nephron development begins in the late stages in embryonic development and proceeds until shortly after birth. Barker and co-workers examined patterns of Lgr5 expression during that time-span in mice. This revealed the existence of a stem-like population of Lgr-positive (Lgr5+ve) epithelial cells localized within primitive nephron precursor structures. The researchers subsequently genetically engineered various strains of mice for a series of 'lineage tracing' experiments, wherein a cell's expression of a gene of interest, such as Lgr5, switches on an indicator gene that will also remain active in that cell's descendants, enabling generation of a visible cellular family tree.
These labeling studies allowed Barker and co-workers to monitor Lgr5+ve cells as they participated in the formation of nephron tubules during kidney development (see image). "We succeeded in demonstrating that these were indeed multipotent, self-renewing stem cells responsible for generating part of the nephron blood filtration unit," says Barker.
Barker is hopeful that these recently discovered stem cells might provide valuable seeds for kidney regeneration in the clinic. "We could try and grow new nephrons in the culture dish or expand these stem cells for use in transplantation into damaged kidneys," he says.
More information: Barker, N., Rookmaaker, M. B., Kujala, P., Ng, A., Leushacke, M., Snippert, H. et al. Lgr5+ve stem/progenitor cells contribute to nephron formation during kidney development. Cell Reports 2, 540–552 (2012). www.cell.com/cell-reports/abstract/S2211-1247%2812%2900258-6