A quantum leap in gene therapy of Duchenne muscular dystrophy
Usually, results from a new study help scientists inch their way toward an answer whether they are battling a health problem or are on the verge of a technological breakthrough. Once in a while, those results give them a giant leap forward. In a preliminary study in a canine model of Duchenne muscular dystrophy (DMD), University of Missouri scientists showed exactly such a leap using gene therapy to treat muscular dystrophy. The results of the study will be published in the journal Molecular Therapy on Jan. 15, 2013.
Muscular dystrophy occurs when damaged muscle tissue is replaced with fibrous, bony or fatty tissue and loses function. Duchenne muscular dystrophy is the most common type of muscular dystrophy predominantly affecting boys. Patients with DMD have a gene mutation that disrupts the production of dystrophin, a protein essential for muscle cell survival and function. Absence of dystrophin starts a chain reaction that eventually leads to muscle cell degeneration and death. For years, scientists have been working to find the key to restoring dystrophin, but they have faced many challenges.
One of the largest hurdles in DMD gene therapy is the large size of the gene. Dystrophin is the largest gene in the human genome, containing approximately 4,000 amino acids. To fit the dystrophin gene into a vehicle that could deliver the gene to the appropriate site in the body, one has to delete 70 percent of the gene. The highly abbreviated gene is known as the "micro-dystrophin" gene. Previous studies suggest that micro-dystrophin can effectively stop muscle disease in mice that are missing dystrophin. However, mice that are missing dystrophin show minimal DMD symptoms, and results from mice often do not predict what will happen in humans. In contrast to mice, loss of dystrophin results in severe muscular dystrophy in dogs. If micro-dystrophin can work in dystrophic dogs, it will likely work in human patients. Unfortunately, when micro-dystrophin was tested in dogs in previous studies, it was not successful.
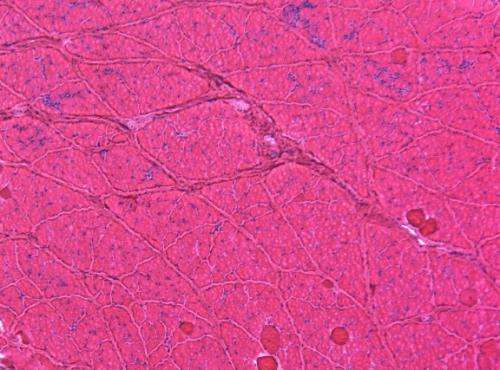
To overcome these hurdles, a team led by Dongsheng Duan, the Margaret Proctor Mulligan Professor in Medical Research at the MU School of Medicine, engineered a new micro-dystrophin gene that carries an important functional region missing in previously tested micro-dystrophins.
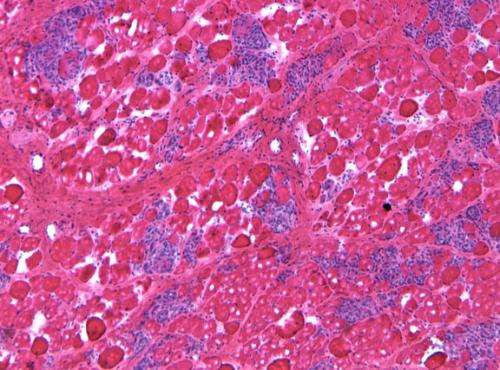
"We placed the new microgene into a virus and then injected the virus into dystrophic dogs' muscles," Duan said. Following gene therapy, Duan's team examined the dogs for signs of muscle disease and measured muscle force in treated and untreated dogs. After careful evaluation of 22 dogs, Duan and colleagues found that the new version of micro-dystrophin not only reduced inflammation and fibrosis, it also effectively improved muscle strength.
"This is the first time that we have seen positive gene therapy results in large mammals of DMD," said Duan. "We still have a lot of work to do, but we now know that our gene therapy strategy works in large mammals; this is a quantum leap forward in fighting this disease. Our next step is to test our strategy in a large group of muscles in the dogs, and then, eventually, see if 'whole body therapy' will work in the dogs. We are still a long way off before we will have a human treatment, but with this finding, I do see a light at the end of this tunnel."
If additional studies, including animal studies, are successful within the next few years, MU officials would request authority from the federal government to begin human drug development (this is commonly referred to as the "investigative new drug" status). After this status has been granted, researchers may conduct human clinical trials with the hope of developing new treatments for Duchenne muscular dystrophy.














