Reassigning cells to fight infection
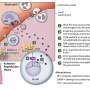
Just as a uniform helps distinguish a soldier from a police officer, scientists use proteins that immune cells wear on their surfaces to determine their job in the body. T cells, for example, that display the CD8 protein are classified as 'cytotoxic lymphocytes', which kill off cancerous or infected cells, whereas those displaying the CD4 protein are identified as 'helper' T cells that coordinate the immune response.
Immunologists have previously viewed these roles as fixed endpoints in development, but research by Ichiro Taniuchi from the RIKEN Research Center for Allergy and Immunology in collaboration with Hilde Cheroutre of the La Jolla Institute for Allergy and Immunology in California and colleagues now reveals that helper T cells retain opportunities for a 'career change'.
Cheroutre had noticed that some helper T cells transplanted into immunodeficient recipients unexpectedly began expressing CD8. "This suggested reprogramming of cells from a helper fate into a cytotoxic lineage," says Taniuchi, whose research group studies ThPOK, a 'master' protein that coordinates helper T cell development and suppresses CD8 production.
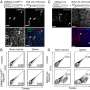
Teaming up to examine these enigmatic CD4+ cytotoxic lymphocytes (CTLs), the groups of Taniuchi and Cheroutre developed genetically modified mice having fluorescently labeled ThPOK-expressing cells, corresponding primarily to helper T cells. They also isolated unlabeled CD4-expressing cells from the intestine, and closer examination showed that these CD4-positive, ThPOK-negative cells now expressed CD8 and behaved like CTLs.
The researchers followed up with a strategy called 'fate mapping', using genetically modified mice whose cells become fluorescently labeled if they express ThPOK at any point—even transiently—in their lifetime. These experiments showed that intestinal CD4+ CTLs begin as ThPOK-expressing helper cells, but switch to a CD8-expressing CTL fate when external factors cause these cells to inactivate ThPOK production via a genetic 'switch' called the 'ThPOK silencer'. "This demonstrates that CD4+ T cells retain unappreciated plasticity for CTL development," says Taniuchi.
This flexibility appears to arise in response to infectious threats, as mice cultivated to be 'germ-free' do not develop CD4+ CTLs. The researchers subsequently determined that helper T cells must be properly exposed both to foreign antigens as well as an immune signaling factor called interleukin-15 to change roles. Although the intestinal wall is a particularly critical point of entry for pathogens, this mechanism may apply more generally as a means of rallying local defenses by retraining helper T cells in a crisis. "I believe other 'barrier sites' in the body may have these cells as well," says Taniuchi.
More information: Mucida, D. et al. Transcriptional reprogramming of mature CD4+ helper T cells generates distinct MHC class II–restricted cytotoxic T lymphocytes. Nature Immunology 14, 281–289 (2013). dx.doi.org/10.1038/ni.2523