Study reveals genes that drive brain cancer
A team of researchers at the Herbert Irving Comprehensive Cancer Center at Columbia University Medical Center has identified 18 new genes responsible for driving glioblastoma multiforme, the most common—and most aggressive—form of brain cancer in adults. The study was published August 5, 2013, in Nature Genetics.
"Cancers rely on driver genes to remain cancers, and driver genes are the best targets for therapy," said Antonio Iavarone, MD, professor of pathology and neurology at Columbia University Medical Center and a principal author of the study.
"Once you know the driver in a particular tumor and you hit it, the cancer collapses. We think our study has identified the vast majority of drivers in glioblastoma, and therefore a list of the most important targets for glioblastoma drug development and the basis for personalized treatment of brain cancer."
Personalized treatment could be a reality soon for about 15 percent of glioblastoma patients, said Anna Lasorella, MD, associate professor of pediatrics and of pathology & cell biology at CUMC.
"This study—together with our study from last year, Research May Lead to New Treatment for Type of Brain Cancer—shows that about 15 percent of glioblastomas are driven by genes that could be targeted with currently available FDA-approved drugs," she said. "There is no reason why these patients couldn't receive these drugs now in clinical trials."
New Bioinformatics Technique Distinguishes Driver Genes from Other Mutations
In any single tumor, hundreds of genes may be mutated, but distinguishing the mutations that drive cancer from mutations that have no effect has been a longstanding problem for researchers.
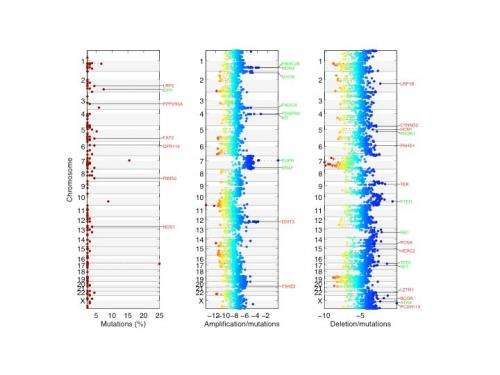
The Columbia team used a combination of high throughput DNA sequencing and a new method of statistical analysis to generate a short list of driver candidates. The massive study of nearly 140 brain tumors sequenced the DNA and RNA of every gene in the tumors to identify all the mutations in each tumor. A statistical algorithm designed by co-author Raul Rabadan, PhD, assistant professor of biomedical informatics and systems biology, was then used to identify the mutations most likely to be driver mutations. The algorithm differs from other techniques to distinguish drivers from other mutations in that it considers not only how often the gene is mutated in different tumors, but also the manner in which it is mutated.
"If one copy of the gene in a tumor is mutated at a single point and the second copy is mutated in a different way, there's a higher probability that the gene is a driver," Dr. Iavarone said.
The analysis identified 15 driver genes that had been previously identified in other studies—confirming the accuracy of the technique—and 18 new driver genes that had never been implicated in glioblastoma.
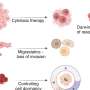
Significantly, some of the most important candidates among the 18 new genes, such as LZTR1 and delta catenin, were confirmed to be driver genes in laboratory studies involving cancer stem cells taken from human tumors and examined in culture, as well as after they had been implanted into mice.
A New Model for Personalized Cancer Treatment
Because patients' tumors are powered by different driver genes, the researchers say that a complicated analysis will be needed for personalized glioblastoma treatment to become a reality. First, all the genes in a patient's tumor must be sequenced and analyzed to identify its driver gene.
"In some tumors it's obvious what the driver is; but in others, it's harder to figure out," said Dr.Iavarone.
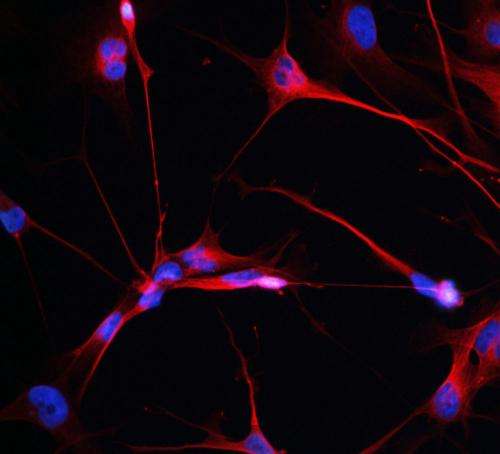
Once the candidate driver is identified, it must be confirmed in laboratory tests with cancer stem cells isolated from the patient's tumor.
"Cancer stem cells are the tumor's most aggressive cells and the critical cellular targets for cancer therapies," said Dr. Lasorella. "Drugs that prove successful in hitting driver genes in cancer stem cells and slowing cancer growth in cell culture and animal models would then be tried in the patient."
Personalized Treatment Already Possible for Some Patients
For 85 percent of the known glioblastoma drivers, no drugs that target them have yet been approved.
But the Columbia team has found that about 15 percent of patients whose tumors are driven by certain gene fusions, FDA-approved drugs that target those drivers are available.
The study found that half of these patients have tumors driven by a fusion between the gene EGFR and one of several other genes. The fusion makes EGFR—a growth factor already implicated in cancer—hyperactive; hyperactive EGFR drives tumor growth in these glioblastomas.
"When this gene fusion is present, tumors become addicted to it—they can't live without it," Dr. Iavarone said. "We think patients with this fusion might benefit from EGFR inhibitors that are already on the market. In our study, when we gave the inhibitors to mice with these human glioblastomas, tumor growth was strongly inhibited."
Other patients have tumors that harbor a fusion of the genes FGFR (fibroblast growth factor receptor) and TACC (transforming acidic coiled-coil), first reported by the Columbia team last year. These patients may benefit from FGFR kinase inhibitors. Preliminary trials of these drugs (for treatment of other forms of cancer) have shown that they have a good safety profile, which should accelerate testing in patients with glioblastoma.
More information: Nature Genetics DOI: 10.1038/ng.2734