December 16, 2013 feature
The brain's got rhythm: Extracting temporal patterns from visual input
(Medical Xpress)—To understand how the brain recognizes speech, appreciates music and performs other higher-level functions, it is necessary to understand how neural systems process temporal information. Recently, scientists at Beijing Normal University studied a simple but powerful network model by which a neural system can extract long-period (several seconds in duration) external rhythms from visual input. Moreover, the study's findings suggest that a large neural network with a scale-free topology – that is, a network in which the probability distribution of the number of connections between its nodes follows a power law – is analogous to a repertoire where neural loops and chains form the mechanism by which exogenous rhythms are learned. Importantly, their model suggests that the brain does not necessarily require an internal clock to acquire and memorize these rhythms.
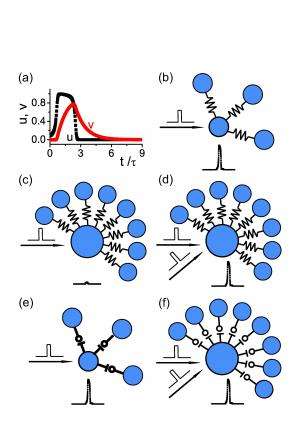
Prof. Si Wu and Prof. Gang Hu discussed the paper that they and their co-authors recently published in Proceedings of the National Academy of Sciences. "The challenge for generating slow oscillation – that is, on the order of seconds – in a neural system is that the dynamics of single neurons and neuronal synapses are too short," Wu tells Medical Xpress. "In other words, for an unstructured network, a strong input will typically generate a strong transient response, and hence the system is unable to retain slow oscillation." To solve this problem, the scientists came up with the idea of using the propagation of activity along a long loop of neurons to hold the rhythm information. "Neurons in the loop need to have low-connectivity degrees to avoid inducing synchronous firing of the network," Hu adds.
Hu also comments on constructing a network model with scale-free structure. "We knew that a scale-free network had the structure we wanted – namely, it consists of a large number of low-degree neurons which can form different sizes of loops and chains, as well as a few hub neurons which can trigger synchronous firing of the network. Furthermore," he continues, "we didn't want hub neurons to be easily elicited; otherwise, the network will always get into epileptic firings." To solve this problem, the researchers required that the neuronal interactions have the proper form to easily activate low-degree neuron while also making it hard to activate hub neurons. Wu point out that biologically plausible electrical synapses and scaled chemical synapses naturally hold this property.
Wu says that the researchers did not develop innovative techniques in this study. "Our main contribution was to propose a simple and yet effective mechanism for a neural system encoding temporal information," he explains, noting that this mechanism consists of five key points:
1. Hub neurons, through their massive connections to others, induce synchronous firing of the network
2. Loops of low-degree neurons hold rhythm information, with the loop size deciding the rhythm
3. Proper electrical or scaled chemical neuronal synapses ensure that activating a hub neuron is difficult in comparison with a low-degree neuron – and also avoids epileptic network firing, in which periods of rapid spiking are followed by quiescent, silent, periods
4. A large-size scale-free network is like a reservoir, which contains a large number and various sizes of loops and chains formed by low-degree neurons, and hence can encode a broad range of rhythmic information
5. When an external rhythmic input is presented, the network selects a loop from its reservoir, with the loop size matching the input rhythm – and this matching operation can be achieved by a synaptic plasticity rule
-

This movie demonstrates that a scale-free network is able to maintain rhythmic synchronous firing. The network has scale-free topology with γ = 3 and mean connectivity k = 4 and consists of N = 210 neurons. Credit: Copyright © PNAS, doi:10.1073/pnas.1304680110 -
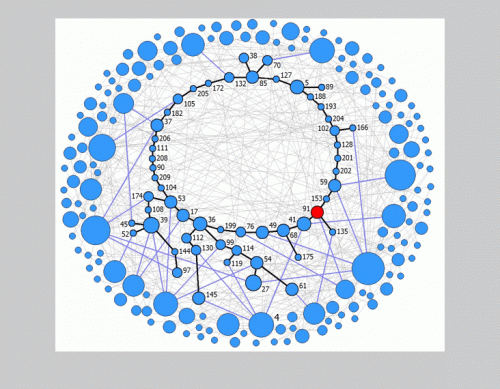
The same network as in Movie S1 but with the neurons’ locations reorganized. The low-degree loop that retains the excitation seed and controls the rhythm of synchronous firing is now located at the center of the figure. Credit: Copyright © PNAS, doi:10.1073/pnas.1304680110
The team's findings imply that in terms of neural information processing, a neural system can use loops and chains of connected neurons to hold the memory trace of input information and, that the latter might serve as the substrate to process temporal events. "These implications for temporal information processing in neural systems have two aspects," Wu points out. "Firstly, there's been a long-standing debate on whether the brain has a global clock that counts time and coordinates temporal events. Our study suggests that this is not necessary: By using intrinsic network dynamics, the neural system can process temporal information in a distributed manner."
Secondly, Wu continues, the brain may not use very complicated strategies to process temporal information, but by fully utilizing its enormous number of neurons, rather simple ones. "Our study suggests that a large size scale-free network has various lengths of loops and chains to hold different rhythms of inputs, making information encoding very simple. This is not economically efficient, but it simplifies computation, which could be crucial for animals responding quickly in a naturally competitive environment."
In the presence of an external rhythmic input, Wu says that the neural system responds and holds the residual activity as the memory trace of the input for a sufficiently long time. If this input is repetitively presented, neuron pairs which fire together become connected through the biological synaptic plasticity rule, and thereby a loop matching the input rhythm is established.
Hu tells Medical Xpress that the network topology is not required to be perfectly scale-free, but rather that the network consists of a few neurons having many connections and a large number of neurons with few connections. "For the convenience of analysis, we considered a scale-free network in which the distribution of neuronal connections satisfying a power law. However, in practice, we don't need such a strong condition. Rather, what we really need is a large number of low-degree neurons forming loops and chains, and a few hub neurons triggering synchronous firing. In other words, scale-free topology is the sufficient, but not the necessary, condition for our model to work." Although the researchers focused on the visual system and have not applied their model to the auditory system, Hi suspects that it can be applied to the latter, where temporal processing is more critical.
Moving forward, the scientists' next step is to build large networks having a similar structure but with more realistic neurons and synapses. "Based on this model," Wu concludes, "we can explore how temporal information encoded in the way proposed in our model is involved in higher brain functions." Moreover, other dynamical systems which generate slow oscillation and need to hold temporal information by network dynamics might benefit from our study."
More information: Long-period rhythmic synchronous firing in a scale-free network, PNAS December 10, 2013 vol. 110 no. 50 E4931-E4936, DOI: 10.1073/pnas.1304680110
© 2013 Medical Xpress. All rights reserved.