January 17, 2014 report
Histamine control of Tourette syndrome
(Medical Xpress)—Like narcolopsy, Tourettes syndrome is as much an enigma to the neuroscientists that study it, as it is to its sufferers. To say that we really understand nothing about how diseases like Tourettes actually work, is to face reality. Yet the standard method of scientific inquiry has finally begun to give us some substantive clues—recently in narcolepsy, and now also in Tourettes. Using an animal model of the disease, researchers have found that one way, among many, to cause the disease, is to create a deficiency of the enzyme histidine decarboxylase (Hdc). Their new findings, just published in Cell, also suggest similar mechanisms may operate in human disease.
When the researchers knocked out the Hdc gene in mice, synthesis of histamine from histadine was impaired, and the mice showed the stereotypical tic behaviors of Tourettes. Regrettably, researchers do no yet know enough about mouse vocalizations to make any generalizations regarding any possible increases in swearing. However, a close parallel with the human symptomatology of Tourettes was indicated by the ability of an infusion of the dopamine D2 receptor antagonist (haloperidol) to fix the tics in the mice. Also, a direct infusion of histamine reduced DA levels in the striatum, which also squares with the current thinking about human Tourettes.
Although haloperidol is a great drug for getting parrots to stop plucking out all their feathers, we can probably do better. The drug has not only been used punitively, for example in the Soviet Union to break prisoners will, but also by our own customs and immigrations enforcement agencies to help smoothen out inconvenient deportations. Today researchers are looking to use more advanced techniques, like deep brain stimulation (DBS), to help reboot brains stuck at odd points in mental-mechanical space.
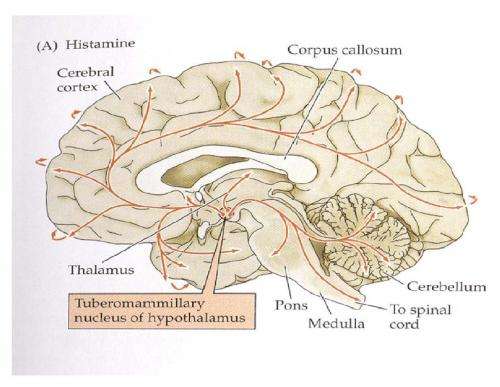
Much of the current effort to treat mental disease through the application of electrical or optical stimulation has been spearheaded by Karl Deiserroth, who has just written an excellent review for Nature in this area. The region that provides histamine for everybody else in the brain is a small area of the posterior hypothalamus called the tuberomammillary nucleus. While it is possible to hit this area precisely using an electrode, the larger effects of any stimulation on the immediate adjacent areas are not so precise.
Stimulation may do more than just excite or inhibit spikes in neurons. If the ultimate result here for tuberomammillary excitation is more histamine release, the hypothesized treatment scenario is that histamine helps keep dopamine under control—whatever control would mean. In some regions, or synaptic specializations of neurons and glia, that would imply more dopamine, in others, perhaps less.
In humans the Hdc "W317X mutation" is rare and therefore only would explain a few cases of Tourettes. As far as what other mechanisms the role of histamine, or for that matter histadine, in Tourettes, nothing is off the table. For example, epigenetic regulation is in now very much invogue and already it has been extended into the therapeutic arena. Regulation of histone acetylation state in the nucleus, by HDAC inhitors, is now seen as a pharmacological target. A recent paper in Cell even suggests a role for HDAC in more esoteric epigenetic phenomena, like memory consolidation. Who is to say that histones, even the ones that are in the slow-turnover pool, are not rate limited by histidine availibility?
In this light, transmitters like histamine might not just be signals that are soon deactivitated or reabsorbed—they can also be envisioned as a metabolic supply, passing either themselves, or their chemical sidegroups off to the next stage. It is interesting that the hypothalamus is implicated in so many mental disorders now, not just Tourettes and Narcolepsy. It is important to gain as much knowledge as possible, particularly in this critical region, before plunging headlong into chemical or electrical stopgaps.
More information: Histidine Decarboxylase Deficiency Causes Tourette Syndrome: Parallel Findings in Humans and Mice, Neuron, Volume 81, Issue 1, 77-90, 8 January 2014, DOI: 10.1016/j.neuron.2013.10.052
Abstract
Tourette syndrome (TS) is characterized by tics, sensorimotor gating deficiencies, and abnormalities of cortico-basal ganglia circuits. A mutation in histidine decarboxylase (Hdc), the key enzyme for the biosynthesis of histamine (HA), has been implicated as a rare genetic cause. Hdc knockout mice exhibited potentiated tic-like stereotypies, recapitulating core phenomenology of TS; these were mitigated by the dopamine (DA) D2 antagonist haloperidol, a proven pharmacotherapy, and by HA infusion into the brain. Prepulse inhibition was impaired in both mice and humans carrying Hdc mutations. HA infusion reduced striatal DA levels; in Hdc knockout mice, striatal DA was increased and the DA-regulated immediate early gene Fos was upregulated. DA D2/D3 receptor binding was altered both in mice and in humans carrying the Hdc mutation. These data confirm histidine decarboxylase deficiency as a rare cause of TS and identify HA-DA interactions in the basal ganglia as an important locus of pathology.
© 2014 Medical Xpress