Possible treatment for kidney disease in lupus studied at UH

In the U.S. alone, more than 1.5 million people have lupus, a chronic autoimmune disease in which the immune system turns against itself, attacking a person's healthy tissue, cells and organs. The symptoms can range from debilitating pain and fatigue to organ failure.
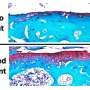
One of the most common organs to be attacked by lupus is the kidney, manifesting in lupus nephritis. An estimated 40 percent of lupus patients develop this condition, which causes inflammation of the kidneys, impairing their ability to get rid of waste products and other toxins from the body effectively. Lupus nephritis is the leading cause of lupus-related deaths and results in tens of thousands of hospitalizations per year.
Dr. Chandra Mohan, a biomedical engineer at the University of Houston (UH), has a theory about the development of lupus nephritis and, if he's right, there may already be a treatment. Mohan was recently awarded a $200,000 grant from the Alliance for Lupus Research (ALR), the world's largest funder of lupus research, to study whether the interaction of three specific molecules is the cause of lupus nephritis. Through this grant, Mohan aims to understand to what extent this molecular pathway is activated in lupus and whether it can be therapeutically targeted using specific drugs.
"Currently, management of lupus involves the use of nonspecific drugs, such as steroids, that suppress the immune system. Our lab, however, is trying to find out the exact molecular mechanisms that lead to lupus," Mohan said. "These mechanisms belong to two classes. On the one hand, you have molecules and pathways that activate the immune system to attack your own body cells. The second set of molecules focus on the kidneys once the immune system is activated by lupus. Our present research and grant focuses on this second molecular cascade, and we want to see whether we can target the kidneys to offer therapeutics that lessen the chance of a patient developing kidney disease once they've developed lupus."
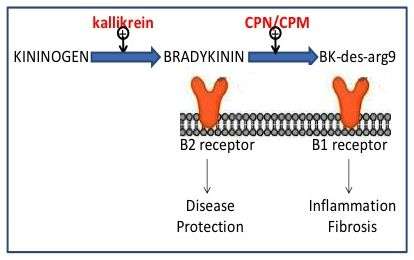
Specifically, Mohan is referring to a pathogenic cascade found in inflammatory kidney diseases, which includes three molecules – kallikreins, bradykinin and one of two bradykinin receptors. High levels of these three specific molecules work together to cause the disease.
The kallikrein enzyme leads to the production of bradykinin, which is a peptide that helps regulate blood pressure and inflammation. The kidneys also have elevated levels of the two bradykinin receptors (B1 and B2), which are proteins that chemically bond with bradykinin to set certain biological processes in motion. While the B2 receptor plays a role in disease protection, the B1 receptor induces inflammation and fibrosis in the kidneys, eventually leading to kidney failure. In lupus patients, B1 is more prevalent. The goal of the ALR grant is to determine whether this pathway can be blocked, thus preventing the activation of certain receptors in the kidneys.
Mohan and his research team, including research assistant professor Yong Du, are working with a drug already in clinical trials for other kidney diseases. This drug acts as an inhibitor that attaches to B1 and physically blocks it from bonding with bradykinin. No bond means no kidney inflammation. Mohan's group will study how subjects with lupus nephritis respond to this inhibitor drug.
"We will compare the efficacy of this novel therapy with that of steroids, which is currently the most common standard of care, in suppressing lupus nephritis," Mohan said. "If these subjects show significant improvement, we will have established the link between lupus nephritis and the pathogenic cascade we are targeting, as well as show that the inhibitor is an excellent candidate for treating lupus nephritis."
Mohan believes there is a high chance this will work, since the company that developed the drug has already gone through many libraries of molecules and selected this inhibitor, which seems to be working in clinical trails with other renal diseases. Additionally, the safety of the inhibitor has already been established, so if his team's experiments go well, he thinks the transition to clinical trials would come quickly.
"To put it in perspective, lupus affects so many pathways and cells. So, if we are successful, this would be one of many different ways of attacking such a complex disease," Mohan said. "We do believe this drug would be more efficacious than the currently available steroids, which essentially dampen the entire immune system."
Mohan, who is a Hugh Roy and Lillie Cranz Cullen Endowed Professor of biomedical engineering in the Cullen College of Engineering, focuses on the genetics of systemic lupus erythematosus (often simply called SLE or lupus). A major goal of his lab is trying to detect who would develop lupus or other chronic rheumatic autoimmune diseases, such as arthritis, and whether individuals will go on to develop kidney, lung, joint, skin or brain diseases, which are all common complications of SLE.