What's Feeding Cancer Cells?
Cancer cells need a lot of nutrients to multiply and survive. While much is understood about how cancer cells use blood sugar to make energy, not much is known about how they get other nutrients. Now, researchers at the Johns Hopkins University School of Medicine have discovered how the Myc cancer-promoting gene uses microRNAs to control the use of glutamine, a major energy source. The results, which shed light on a new angle of cancer that might help scientists figure out a way to stop the disease, appear Feb. 15 online at Nature.
“While we were looking for how Myc promotes cancer growth, it was unexpected to find that Myc can increase use of glutamine by cancer cells,” says Chi V. Dang, M.D., Ph.D., the Johns Hopkins Family Professor of Oncology at Johns Hopkins. “This surprising discovery only came about after scientists from several disciplines came together across Hopkins to collaborate — it was a real team effort.”
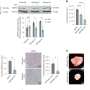
In their search to learn how Myc promotes cancer, the researchers teamed up with protein experts, and using human cancer cells with Myc turned on or off, they looked for proteins in the cell’s powerhouse — the mitochondria — that appeared to respond to Myc. They found eight proteins that were distinctly turned up in response to Myc.
At the top of the list of mitochondrial proteins that respond to Myc was glutaminase, or GLS, which, according to Dang, is the first enzyme that processes glutamine and feeds chemical reactions that make cellular energy. So the team then asked if removing GLS could stop or slow cancer cell growth. Compared to cancer cells with GLS, those lacking GLS grew much slower, which led the team to conclude that yes, GLS does affect cell growth stimulated by Myc.
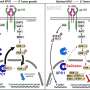
The researchers then wanted to figure out how Myc enhances GLS protein expression. Because Myc can control and turn on genes, the team guessed that Myc might directly turn on the GLS gene, but they found that wasn’t the case. “So then we thought, maybe there’s an intermediary, maybe Myc controls something that in turn controls GLS,” says Ping Gao, Ph.D., a research associate in hematology at Johns Hopkins.
They then built on previous work done with the McKusick-Nathans Institute of Genetic Medicine at Hopkins where they discovered that Myc turns down some microRNAs, small bits of RNA that can bind to and inhibit RNAs, which contain instructions for making proteins. The team looked more carefully at the GLS RNA and found that it could be bound and regulated by two microRNAs, called miR23a and miR23b, pointing to the microRNAs as the intermediary that links Myc to GLS expression.
“Next we want to study GLS in mice to see if removing it can slow or stop cancer growth,” says Gao. “If we know how cancer cells differ from normal cells in how they make energy and use nutrients, we can identify new pathways to target for designing drugs with fewer side effects.”
This study was funded by the National Institutes of Health, the National Cancer Institute, the Rita Allen Foundation, the Leukemia and Lymphoma Society and the Sol Goldman Center for Pancreatic Cancer Research.
Authors on the paper are Ping Gao, Irina Tchernyshyov, Tsung-Cheng Chang, Yun-Sil Lee, Karen Zeller, Angelo De Marzo, Jennifer Van Eyk, Joshua Mendell and Chi V. Dang, of Johns Hopkins; and Kayoko Kita and Takfumi Ochi of Teikyo University in Japan.
On the Web:http://www.nature.com/nature/index.html
Provided by Johns Hopkins University School of Medicine