This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Team finds exportin 1 governs immunosuppressive functions of myeloid-derived suppressor cells in tumors
A team of Roswell Park Comprehensive Cancer Center experts led by Hemn Mohammadpour, DVM, Ph.D., has conducted research that offers new insights into tumor biology and may lay the groundwork for more effective cancer immunotherapy.
Their preclinical findings were published June 20 in the journal Cellular & Molecular Immunology.
"We discovered that a protein highly expressed by tumor cells and targeted in treatments for cancers like multiple myeloma is also highly expressed by myeloid-derived suppressor cells," notes Saeed Daneshmandi, Ph.D., study first author and a postdoctoral researcher with the Department of Cell Stress Biology at Roswell Park.
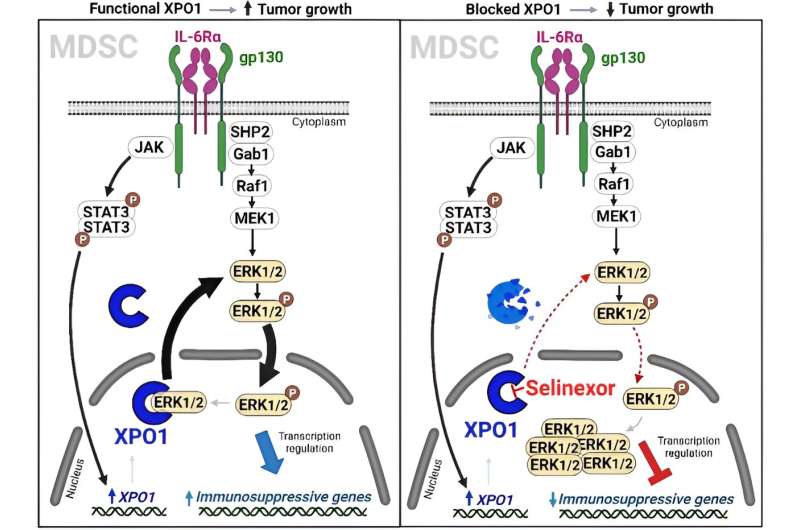
"This discovery is important because it reveals the previously unknown immunoregulatory mechanism of the FDA-approved Exportin 1 inhibitor selinexor, suggesting that XPO1 blockade could be effective in combination therapy—particularly in solid tumors, where selinexor alone has shown limited success."
Myeloid-derived suppressor cells (MDSCs) are known to promote tumor progression through the suppression of the body's antitumor immune response. Gaining further understanding into the mechanisms that govern their development and immunosuppressive functions could reveal new therapeutic targets to improve antitumor immunity.
Exportin 1 (XPO1), a nuclear export protein, plays an essential part in the transport of many proteins and RNA molecules from the nucleus to the cytoplasm. Overexpression of XPO1 is common in many cancer types, including multiple myeloma, pancreatic, ovarian, glioma, lung, stomach, prostate and colorectal tumors, and is associated with poor prognosis.
In a prior exploratory single-cell RNA sequencing study published in Nature Communications that explored MDSC heterogeneity, Dr. Mohammadpour and colleagues uncovered the potential importance of XPO1 for myeloid-derived suppressor cell differentiation and function.
Despite extensive knowledge regarding XPO1's role in cancer cells, its function in myeloid cells, particularly myeloid-derived suppressor cells, remains largely unexplored. Dr. Mohammadpour and colleagues aimed to investigate XPO1's impact on MDSC differentiation and immunosuppressive functions, as well as to understand the mechanisms by which XPO1 regulates these properties in MDSCs.
"Our findings revealed that selinexor, an FDA-approved XPO1 inhibitor, reduces tumor progression partly by blocking MDSCs, inducing neutrophil-like cells with antitumor capabilities, and enhancing the antitumor immune response," says Dr. Mohammadpour.
"This discovery is significant because it suggests that selinexor could be used as an immunomodulatory agent to improve the effectiveness of standard treatments, such as immune checkpoint inhibition."
"The potential benefit of combining XPO1 blockade with existing and future immunotherapies holds promise for improving patient outcomes while also expanding the arsenal of available treatment options for patients with various cancer types," Dr. Mohammadpour says.
More information: Saeed Daneshmandi et al, Exportin 1 governs the immunosuppressive functions of myeloid-derived suppressor cells in tumors through ERK1/2 nuclear export, Cellular & Molecular Immunology (2024). DOI: 10.1038/s41423-024-01187-1
Saeed Daneshmandi et al, Myeloid-derived suppressor cell mitochondrial fitness governs chemotherapeutic efficacy in hematologic malignancies, Nature Communications (2024). DOI: 10.1038/s41467-024-47096-9