Signaling pathway reveals mechanism for B cell differentiation in immune response
The finding establishes a role for the extracellular signal-regulated kinase (ERK) signaling pathway in B cell differentiation, a key step toward the development of B cell-targeted drugs for treatment of autoimmune diseases and allergies.
As the only cells in the body that produce antibodies, B cells play an essential role in the immune system’s defense against bacteria and viruses. Differentiation of B cells into antibody-producing plasma cells is central to this role, initiating the production of antibodies whose targeted binding mechanism enables the immune system to identify and neutralize foreign objects. The mechanism underlying this differentiation process, however, remains unknown.
To better understand this mechanism, the research group focused on the signaling of the extracellular signal-regulated kinases (ERK), intracellular signaling molecules known to play an important role in the cell cycle and survival of immune cells. Hoping to glean insights into the role of ERKs in B cell differentiation into plasma cells, the researchers generated mice deficient in two different ERKs, ERK1 and ERK2, and studied the effect of this deficiency on the fate of B cells.
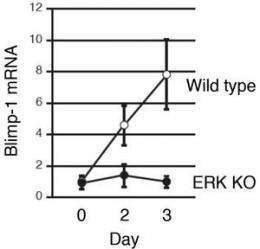
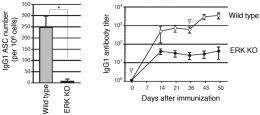
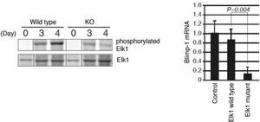
What they found confirmed that ERKs are in fact essential to B cell differentiation: B cells in mice without these key molecules were unable to form plasma cells (Figure 1). The researchers further traced this observation to a gene called Prdm1 encoding the protein BLIMP-1, increased expression of which leads to differentiation and proliferation of plasma cells in B cell immune response (Figure 2). ERKs, they discovered, phosphorylate the transcription factor Elk1, which leads to expression of Blimp-1 (Figure 3).
By elucidating the role of ERKs in B cell differentiation, the current research provides valuable insight into a little-understood area of immune response, promising advances in drug discovery and offering hope to autoimmune disease and allergy sufferers around the world.
More information: T. Yasuda, et al., ERKs induce expression of the transcriptional repressor Blimp-1 and subsequent plasma cell differentiation. Sci. Signal. 4, ra25 (2011). DOI: 10.1126/scisignal.2001592














