The tangled NETs of the immune system
When scientists can’t believe their eyes, it is very likely that they are on to something quite extraordinary. This was precisely the case for Arturo Zychlinsky and his colleagues at the Max Planck Institute for Infection Biology in Berlin. White blood cells that cast net-like structures to ensnare pathogens? No one had ever seen the likes of it before. Now the first patients are reaping the benefits of this discovery.
The boy could hardly breathe and required oxygen support. He found it very difficult to walk. The fungal infection in his lungs was threatening to spread. The eight-year-old had suffered from an immune system disorder known as chronic granulomatous disease (CGD) since birth. He had been laid low by recurring fungal infections and nobody knew why his immune system found it difficult to fight these particular invaders.
A gene therapy developed by a team headed by Janine Reichenbach and Rein hard Seger at Zurich University Hospital saved the boy’s life. “It’s wonderful,” enthuses Volker Brinkmann, leader of the Microscopy Research Group at the Max Planck Institute for Infection Biology in Berlin. The biologist was involved only indirectly in the treatment of the young patient – or, to be more precise, his contribution was purely scientific. It was based on his research on neutrophil granulocytes, cells that play a very important role in the innate immune system, and on the netlike structures these cells create to ensnare pathogens – Neutrophil Extracellular Traps, NETs for short.
One of the most exciting immunological discoveries of recent decades comes from the institute located on the site of the famous Charité Hospital in Berlin, once the workplace of legendary infectious disease researchers Robert Koch and Paul Ehrlich. Based on the studies carried out by the Berlin-based scientists, it became evident that the young patient’s neutrophils were incapable of forming NETs. According to Brinkmann, this “has dire consequences, particularly in the fight against fungal infections.”
An artificial or natural phenomenon?
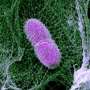
The scientist is in his office examining an image of the NETs, taken using one of his high-tech microscopes in the adjacent laboratories. From a purely aesthetic perspective, the image is nothing short of delightful and resembles, at first glance, a cluster of vividly colored threads and dots. Brinkmann was the first person to see something similar to this image in 2003, when nobody knew of the existence of these defensive cellular and molecular webs that suddenly appeared under his microscope.
Arturo Zychlinsky, Director at the Max Planck Institute for Infection Biology and head of the Cellular Microbiology research group, was also initially at a loss when presented with these strange structures. “I thought it was an artifact,” he admits – in other words, an artificial phenomenon that is generated by laboratory conditions and does not arise naturally in the organism.
What’s more, Arturo Zychlinsky, who was born and bred in Mexico City, had a very long track record in infection biology and was a renowned expert in the field. As a young man he was fascinated by microbes and the immune system that defends the body against them. It was thus clear to him from an early age that the study of microbiology and immunology was the only career option worth pursuing. When it eventually came to choosing a topic for his doctorate, he opted for a subject relating to innate immune defense.
Compared to the sophistication of the acquired or adaptive immune system, the innate immune system was considered a rather simple and blunt instrument. It is based on the principle of using tailor-made weapons to destroy all germs, be they viral, bacterial, parasitical or fungal in nature, and thereafter creating a long-term memory of the pathogen’s molecular calling card to enable faster and more effective action in the case of a repeat infection.
The adaptive immune system consists of a myriad of specialized cells with various sub-classes, even more messenger substances and signaling molecules, and tailor-made antibodies – in other words, a veritable and inconceivably complex jungle of cellular communication and signal processing. In the young Zychlinsky’s view, it was “too complicated for me to research,” as he candidly admits.
The supposedly less complex innate immune system presented a far more attractive proposition. Even the ancient Greeks knew of its existence and described inflammatory processes. “Inflammation is the response of the innate immune system to infection,” says Zychlinsky, and involves, among other things, pain and the reddening of tissue. The innate immune response is a kind of vanguard, an initial firewall that attacks invading pathogens in a matter of minutes and, superficially at least, involves a less intricate line of defense than the adaptive immune system.
“It’s as old as the hills,” speculates Zychlinsky with good reason; probably as old as multicellular animal life itself, so a good 500 to 600 million years old. Its cells and molecules have existed in sponges and insects, fish and frogs since organisms started to live off and exploit other organisms.
Fast-reacting innate immune system
The macrophages are among the most important cells of the innate immune system. They literally devour germs – albeit with far less gusto than the neutrophil granulocytes that fulfill this task a hundred times more effectively and that the bone marrow produces daily by the millions. Of all white blood cells, 70 percent are neutrophils. And adult humans are fortunate enough to carry a good half pound of them around in their systems. They roam through the blood and simply await activation by the immune system’s custodian cells (the dendritic cells), which patrol the body’s tissues and are activated in turn by messenger substances like interleukins.
When they receive the signal “pathogens found in neighboring tissue!”, the rounded neutrophils, with their extensively lobed cell nucleus, rush from the blood stream to the wall of the vessel in which they are currently located, seek out the tiniest opening, make themselves as elongated and slim as possible, slip through the opening, and hasten to the site of the infection.
“These foot soldiers of the immune system,” as Arturo Zychlinsky calls them, are very well armed and always primed for action. They do not divide and, irrespective of whether they have been deployed or not, die after a maximum lifespan of six hours. Once they reach the site of the bacterial invasion, they decide which approach to adopt against the invader. They either devour it in a split-second process or they flood the infected area with granule proteins, bacteria killers they have stored in their dozens of small depots. “These contain a top-class arsenal for fighting pathogens,” explains Volker Brinkmann.
One of these enzymes is neutrophil elastase, which Arturo Zychlinsky’s team has been investigating since his time at New York University School of Medicine. Elastase also annihilates the bacterial proteins that render the microbes pathogenic to humans. Zychlinsky and his colleagues have shown that elastase can break down the virulence factors of Shigella bacteria, which cause dysentery, a dangerous diarrheal illness.
In 2003, the Berlin researchers wanted to identify the place where the Shigella bacteria and elastase meet in the neutrophils. This was a task for Volker Brinkmann and his microscopy team, who used a stain to locate the elastase in a neutrophil culture that had been infected with Shigella, enabling them to make it visible under their light and electron microscopes. “Elastase suddenly appeared in these colorful balls located outside the cells,” remembers Brinkmann. “That was strange.”
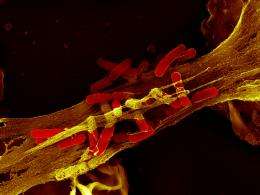
The scientists then conducted further experiments in which they also deliberately stained the neutrophils’ genetic material. This sophisticated approach provided definitive proof of the existence of the NETs: “They were always there,” says Brinkmann. The possibility that they were an artifact could thus be excluded.
Chromatin in the nets acts as a potent antibiotic
Since publication of this astonishing discovery in the journal Science, numerous research teams have tilled this new field, including, of course, the Berlin-based scientists. Today, seven years later, they have an impressive film to show their guests, presenting the process in fast motion. It is a highly dynamic process accompanied by dramatic changes in the neutrophil granulocytes. “Like suicide bombers who blow themselves up,” is how doctoral student Kathleen Metzler describes the scene, since the formation of the NETs also signals the simultaneous demise of the granulocytes.
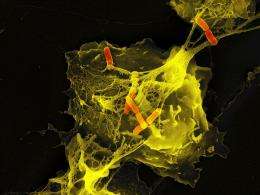
Under the scanning electron microscope, the NETs appear as fine fibers and particles that link the threads to form more complex structures. This causes the formation of a ball in which the bacteria become engulfed. The main ingredient of this ball is chromatin. This mixture of DNA and proteins is normally found in the cell nucleus and contains genetic information.
Most of the proteins contained in the chromatin are histones, which lend the DNA an ordered structure. During the formation of the NETs, some of the histones are cut so that the DNA threads can unfold. In addition to chromatin and histones, the NETs also contain between 20 and 25 proteins from the cell plasma of the neutrophils that can kill the bacteria or render their virulence factors harmless.
In Arturo Zychlinsky’s view, however, the histones are “the most important antimicrobial substances in the NETs.” The fact that histones can fight bacteria was discovered by US scientist James Hirsch as far back as 1958. His finding, however, did not attract much support among immunology experts. Histones that have been purified in the laboratory are far more effective at killing bacteria than all other known autologous antibiotics. “But we have absolutely no idea how they do it,” says the Max Planck researcher.
It is also unclear whether histones in the organism play a role in the immune response. Zychlinsky suspects that the histones developed in the course of evolution to protect genetic material from bacterial attacks. However, he is unable to prove this. What is known is that the fatal NETs ensnare fungi and, of course, bacteria, such as Shigella, Salmonella, pneumococci and staphylococci, which can cause food poisoning and toxic shock syndrome.
Neutrophils sacrifice themselves
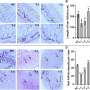
The Max Planck scientists demonstrated the presence of NETs not only in experimental cell cultures but also in tissue samples from patients with appendicitis and bacillary dysentery (shigellosis). Interestingly, this process functions just as well as the devouring of the pathogens: “As many bacteria perish in the NETs as are digested by living neutrophils,” says Arturo Zychlinsky.
The aforementioned film also shows how the neutrophils can mobilize vital components of the cell nucleus and eject them from the cell. Once activated by the presence of a bacterium in the vicinity or by immune system messenger substances, the cells run a program that inevitably signals their own demise – a case of suicide for the common good. Initially, the structure of the nucleus and the granules changes. “The membrane around the nucleus disintegrates, the granules dissolve, and the components of the NETs can mix in the cell interior as a result,” says Volker Brinkmann.
All of this unfolds within a period of two hours. A stage between life and death follows. In the final act, after three hours, the cell contracts again until the cell membrane tears and the highly active mixture is expelled. Outside the cell, it unfolds and forms the NETs, which engulf the bacteria.
“We do not yet know what happens at the molecular level,” says Zychlinsky. Due to their short lifespan, neutrophils are very difficult to study in the laboratory. What is definite is that when certain receptors on the neutrophil surface receive the death signal, within 20 minutes, the cell activates an enzyme known as NADPH oxidase that produces aggressive oxygen radicals from normal oxygen molecules – so-called superoxide. When the Berlin researchers inhibit this enzyme, NET formation does not occur.
Kathleen Metzler worked with chronic granulomatosis disease patients, fellow sufferers of the eight-year-old gene therapy patient. The genes for the production of NADPH oxidase are defective in these patients, so they are unable to form NETs. As the American scientist discovered, people with a mutation in the gene for the enzyme myeloperoxidase – which is also involved in oxygen radical formation – are also unable to form NETs. However, the resulting immune weakness is often less serious in this case. Using cell samples from these patients, the doctoral student and her colleagues aimed to establish why oxygen radicals are so crucial to NETs formation.
NETs degradation is distorted in Lupus patients
Like every useful biological phenomenon, the formation of the NETs comes at a price. The Max Planck researchers suspected this from the outset and referred to a serious autoimmune disease, systemic lupus erythematosus (SLE), in their original publication in the journal Science. With autoimmune diseases, the adaptive immune system incorrectly directs its weapons, for example antibodies, at structures belonging to its own organism.
Lupus is ten times more common in women than in men. Its characteristic symptoms, which include skin rashes – such as the typical butterfly-shaped facial redness – joint and muscle pain, fatigue, and inflammation, are probably triggered by infections. Lupus progresses in bouts known as flares, and gets worse with each progressive flare. In some patients, the autoimmune antibodies destroy the kidneys.
A lupus diagnosis, which is difficult to make, depends on numerous factors. In all cases, the patients form three types of antibodies: against the genetic substance DNA, against histones, and against proteins from the neutrophil granules. Hence, interestingly, it produces antibodies against the specific components of the NETs. Moreover, an SLE attack often occurs after an infection. The scientists in Berlin thus suspect that the symptoms may be caused by the failure of the NETs to disintegrate.
Together with medical researchers from the University of Erlangen, the Max Planck researchers went to the bottom of the matter. With each new lupus flare following an infection, neutrophils apparently migrate to the center of the action and produce NETs. The timely degradation of the NETs appears to be essential to prevent the formation of new autoimmune antibodies. It would appear that the enzyme DNase-1, which circulates in the blood, usually degrades the NETs. However, this process does not work in some lupus patients. “Either they form too many NETs or the DNase is blocked,” says Arturo Zychlinsky, perhaps by antibodies against the enzyme. Or the DNase-1 simply does not reach the NETs.
As the scientists have discovered, when this mechanism is defective, the antibodies accumulate particularly in the kidneys. “We can say with a considerable degree of certainty that these lupus patients have a high risk of kidney failure,” stresses Zychlinsky. Based on this discovery, it may be possible to develop a new diagnostic test to enable the prognosis of kidney failure based on the presence of an elevated concentration of antibodies against the NETs. This could also open the door to the earlier treatment of affected patients.
But there is still a long way to go before a treatment is developed for lupus patients. One possibility is to filter the dangerous antibodies using a kind of dialysis process. However, if the scientists succeed in decoding the molecular secrets of the disease process, a selectively effective drug may one day be found that can stop the formation of NETs in SLE patients.
NETs play a role in vasculitis, pre-eclampsia and cystic fibrosis
The NETs also play an important role in the emergence of another rare autoimmune disease – vasculitis – in which the immune system attacks the blood vessels and kidneys. Scientists are discovering more and more that NETs are involved in disease processes – or they are suddenly able to explain previously mysterious phenomena using NETs. For instance, the reason why bacteria known as staphylococci and pneumococci place a DNase on their surface had long mystified scientists. Following a research study carried out by the Max Planck researchers in Berlin and scientists at the Karolinska Institute in Stockholm, the reason for this is now clear: the purpose of the enzyme is to hack away at the NETs. If the DNases are removed from the pathogens, they pose far less of a threat to the organism. An experiment on mice has proven that, in the absence of DNases, the bacteria do not migrate to the lungs, and the risk of blood poisoning, for example, is practically eliminated.
Moreover, the NETs are clearly involved in the emergence of pre-eclampsia in pregnancy. This disease causes pregnant women to store water in their tissues and develop high blood pressure. The process can also have very serious consequences for the unborn child. The external layer of the maternal tissue forms particles in the area of contact with the embryo. If an inflammation occurs and the neutrophils arrive, NETs are formed that threaten the blood supply to the embryo and that actually “congeal” the tissue, as Volker Brinkmann explains. This deprives the embryo of oxygen, with the associated potentially dramatic consequences.
Researchers working with Dominik Hartl at Munich University Hospital also found masses of NETs in the lungs of patients with cystic fibrosis. Patients with this disease suffer from congestion of the lungs – fertile ground for recurring bacterial infections – as a result of a genetic defect. The NETs also block the lungs and exacerbate the patients’ breathing problems. The worse the patients’ pulmonary function is, the more NETs they have in their airways. In this instance, it would appear that NET formation is dependent on NADPH oxidase.
According to the findings of former colleagues of Arturo Zychlinsky, NETs stimulate the formation of blood clots in veins and provide an ideal framework for them. Blood platelets adhere to the NETs, are activated and form clumps. The administration of DNase halts this dangerous process. The NETs also connect with numerous proteins that promote thrombosis. The drug heparin can also halt the formation of thromboses triggered by the NETs. The reason for this is that heparin removes the histones from the DNA. It appears that histones alone can cause the formation of the blood clots. These findings confirm an observation made in the context of population studies whereby infections are frequently linked with the formation of blood clots.
This shows how the ripples created by Arturo Zychlinsky and Volker Brinkmann’s discovery, made with the help of savvy, skill and chance, continue to spread. “We are interested above all in basic scientific processes,” stresses Zychlinsky. “And if a patient benefits at some point, it would make us the happiest people on earth.”