Cancer paradigm shift: Biomarker links clinical outcome with new model of lethal tumor metabolism
Researchers at the Kimmel Cancer Center at Jefferson have demonstrated for the first time that the metabolic biomarker MCT4 directly links clinical outcomes with a new model of tumor metabolism that has patients "feeding" their cancer cells. Their findings were published online March 15 in Cell Cycle.
To validate the prognostic value of the biomarker, a research team led by Agnieszka K. Witkiewicz, M.D., Associate Professor of Pathology, Anatomy and Cell Biology at Thomas Jefferson University, and Michael P. Lisanti, M.D., Ph.D., Professor and Chair of Stem Cell Biology and Regenerative Medicine at Jefferson, analyzed samples of patients with triple negative breast cancer, one of the most deadly of breast cancers, with fast-growing tumors that often affect younger women.
A retrospective analysis of over 180 women revealed that high levels of the biomarker MCT4, or monocarboxylate transporter 4, were strictly correlated with a loss of caveolin-1 (Cav-1), a known marker of early tumor recurrence and metastasis in several cancers, including prostate and breast.
"The whole idea is that MCT4 is a metabolic marker for a new model of tumor metabolism and that patients with this type of metabolism are feeding their cancer cells. It is lethal and resistant to current therapy," Dr. Lisanti said. "The importance of this discovery is that MCT4, for the first time, directly links clinical outcome with tumor metabolism, allowing us to develop new more effective anti-cancer drugs."
Analyzing the human breast cancer samples, the team found that women with high levels of stromal MCT4 and a loss of stromal Cav-1 had poorer overall survival, consistent with a higher risk for recurrence and metastasis, and treatment failure.
Applying to a Triple Threat
Today, no such markers are applied in care of triple negative breast cancer, and as a result, patients are all treated the same. Identifying patients who are at high risk of failing standard chemotherapy and poorer outcomes could help direct them sooner to clinical trials exploring new treatments, which could ultimately improve survival.
"The idea is to combine these two biomarkers, and stratify this patient population to provide better personalized cancer care," said Dr. Witkiewicz.
The findings suggest that when used in conjunction with the stromal Cav-1 biomarker, which the authors point out has been independently validated by six other groups worldwide, MCT4 can further stratify the intermediate-risk group into high and low risk.
Since MCT4 is a new druggable target, researchers also suggest that MCT4 inhibitors should be developed for treatment of aggressive breast cancers, and possibly other types. Targeting patients with an MCT4 inhibitor, or even simple antioxidants, may help treat high-risk patients, who otherwise may not respond positively to conventional treatment, the researchers suggest.
Paradigm Shift
But the work stems beyond triple negative breast cancer, challenging an 85-year-old theory about cancer growth and progression.
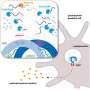
This paper is the missing clinical proof for the paradigm shift from the "old cancer theory" to the "new cancer theory," known as the "Reverse Warburg Effect," said Dr. Lisanti. The new theory being that aerobic glycolysis actually takes place in tumor associated fibroblasts, and not in cancer cells, as the old theory posits.
"The results by Witkiewicz et al. have prominent conceptual and therapeutic implications," wrote Lorenzo Galluzzi, Ph.D., Oliver Kepp, Ph.D., and Guido Kroemer, M.D., Ph.D. of the French National Institute of Health and Medical Research and Institut Gustave Roussy, in an accompanying editorial. "First, they strengthen the notion that cancer is not a cell-autonomous disease, as they unravel that alterations of the tumor stroma may constitute clinically useful biomarkers".
"Second, they provide deep insights into a metabolic crosstalk between tumor cells and their stroma that may be targeted by a new class of anticancer agents."
Dr. Kroemer entitled his commentary "Reverse Warburg: Straight to Cancer" to emphasize that the connective tissue cells (fibroblasts) are directly "feeding" cancer cells, giving them a clear growth and survival advantage. New personalized therapies would cut off the "fuel supply" to cancer cells, halting tumor growth and metastasis.