New study should end debate over magnesium treatment for preventing poor outcome after haemorrhagic stroke
An international randomised trial and meta-analysis published Online First in The Lancet should put an end to the debate about the use of intravenous magnesium sulphate to prevent poor outcomes after haemorrhagic stroke. The investigators found no benefit from magnesium treatment compared with placebo.
"The findings from MASH 2 have important implications for clinical practice. Administration of magnesium is standard practice in many centres", explains Sanne Dorhout Mees from University Medical Center Utrecht, Netherlands, who led the research.
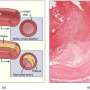
About a third of patients with aneurysmal subarachnoid haemorrhage (SAH; the bursting of a blood vessel on the surface of the brain), the deadliest form of stroke, will die within the first month. Of those that survive, 20% are left disabled. Delayed cerebral ischaemia can occur 4 to 10 days after SAH and is an important cause of poor outcome.
The benefits of intravenous magnesium sulphate to limit the damage from SAH are controversial. Despite promising findings from earlier research, more recent studies have reported conflicting results.
Here, the investigators report the results of a randomised trial comparing magnesium treatment (606 patients) with placebo (saline; 597 patients) in adults with SAH from the Netherlands, Scotland, and Chile. The main outcome measure was poor outcome defined as a score of 4 to 5 on the modified Rankin Scale (meaning severe disability), or death, 3 months after haemorrhage.
No difference in poor outcome was recorded between the groups at 3 months—158 patients (26.2%) given magnesium compared with 151 (25.3%) given placebo had poor outcome.
A further meta-analysis of seven randomised trials involving over 2000 patients with SAH found magnesium was not superior to placebo.
The authors conclude: "Results from MASH 2 alone, and in combination with the other trial data, show that intravenous magnesium does not affect outcome after aneurysmal subarachnoid haemorrhage. Subgroup analyses did not identify a subgroup of patients who might benefit from magnesium treatment…therefore routine administration of magnesium cannot be recommended."
In a linked Comment, Chethan Venkatasubba Rao and Jose Suarez from Baylor College of Medicine, Texas, USA remark: "The results, although disappointing, are nonetheless very pertinent to the management of patients with aneurysmal subarachnoid haemorrhage. We agree with the researchers, and previous findings, that routine intravenous magnesium sulphate infusion cannot be recommended for patients with aneurysmal subarachnoid haemorrhage."
More information: Paper online: www.thelancet.com/journals/lan … (12)60724-7/abstract