New medicine might fight drug-resistant TB, study says
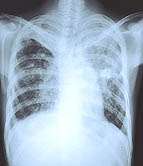
(HealthDay) -- More than four decades after the last introduction of fresh treatments for tuberculosis, a new medication for multidrug-resistant strains of the global scourge has shown promise in a multinational trial.
Scientists in nine countries tested delamanid -- which inhibits the production of mycolic acid, a key component of tuberculosis (TB) bacteria. In a phase 2 trial with 481 patients, the drug cleared TB from the sputum cultures of nearly half the patients within two months, the study said.
"I think it's very important that we have, in delamanid, the potential for a new drug in the first class of new drugs in 40 years," said study co-author Lawrence Geiter, vice president of global clinical development for Otsuka Pharmaceutical Co., the Tokyo-based developer of delamanid. "It's going to enhance treatment options."
The study is published June 7 in the New England Journal of Medicine.
Considered one of the world's deadliest infectious diseases, TB or its complications kill about 1.5 million people globally each year. The World Health Organization (WHO) estimates that nearly 5 percent of all TB cases worldwide are multidrug-resistant, with 440,000 cases occurring annually. If first-line drugs such as rifampin and isoniazid fail, second-line drugs must be taken for up to two years, and a cure is far from guaranteed.
Though TB rates fell to an all-time low in the United States in 2011, the U.S. Centers for Disease Control and Prevention found that infection rates were seven times higher for Hispanics, eight times higher for blacks and 25 times higher for Asians than whites.
"Any time you can add a new TB drug with a novel mechanism that kills bacteria, it's an important advance," said Dr. Eric Nuermberger, an associate professor of medicine at the Center for Tuberculosis Research at Johns Hopkins University School of Medicine, in Baltimore. "Drug-resistant TB is becoming a huge global crisis, and this study proves that this drug . . . has the potential to improve our current regimens. We can all sit and speculate about the margin of improvement, but this proves that it's active nonetheless."
Study participants, who were between 18 and 64 years old, were randomly assigned to one of three equal groups, including two groups that were administered different daily doses of delamanid and a third that received a placebo. All three groups also took a background drug regimen developed according to WHO guidelines.
After eight weeks of treatment, about 45 percent of patients receiving 100 milligrams (mg) of delamanid twice daily were clear of TB bacteria in sputum cultures, as were nearly 42 percent of those receiving 200 mg of delamanid twice daily. This rate was only about 30 percent for those in the placebo group.
A potentially concerning side effect of delamanid treatment was a change in the heart's electrical activity known as QT prolongation, which showed up on electrocardiograms but did not produce cardiac symptoms such as dizziness or heart arrhythmias. Geiter said phase 3 studies of the drug will continue to monitor the effect. Otsuka has applied for drug approval in the European Union but not yet in the United States, he added.
Nuermberger noted that several new TB drugs under development have the potential to affect the heart, "so the real concern is about the additive effects" should these drugs be used in combination with each other.
"Those things are going to have to be sorted out," Nuermberger said. "These are the types of problems that don't tend to show up in a phase 2 study . . . even when they are effects on the EKG, as described here, death and arrhythmias are relatively infrequent. But when you apply them on a global basis, it can become important."
"While it's a critical advance, there still a tremendous amount of work that needs to be done to combine [delamanid] with existing agents . . . and turn the tide," added Nuermberger, who co-authored a commentary on treatments for multidrug-resistant TB in the same journal issue.
More information: The U.S. Centers for Disease Control and Prevention has more about multidrug-resistant TB.
Copyright © 2012 HealthDay. All rights reserved.