Starr collaboration illuminates mysterious pathway to immortality in cancer cells
(Medical Xpress) -- Cancer cells are immortal because they circumvent failsafe mechanisms that stop out-of-control cell proliferation. One of these mechanisms – the progressive shortening of chromosomes – is prevented by replenishment of telomeres, the protective elements at the ends of chromosomes. Most cancer cells do this with an enzyme called telomerase, but approximately 10 to 15 percent of human cancers use a different pathway called ALT (alternative lengthening of telomeres). Whether or not a tumor has ALT makes a difference. For example, people with glioblastoma live twice as long if the tumor uses ALT.
With support from the Starr Cancer Consortium, a team of scientists from The Rockefeller University, The Broad Institute of MIT and Harvard, Memorial Sloan-Kettering Cancer Center and Weill Cornell Medical College set out to demystify ALT. The team carried out a detailed analysis of a large panel of ALT cell lines for their genetic and cell biological features. How ALT is activated was not known but recent data pointed the finger at an enzyme called ATRX. ATRX changes the way DNA wraps around proteins in chromosomes.
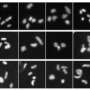
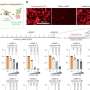
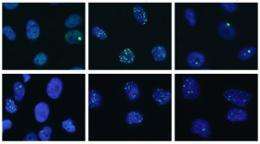
In an article published online July 19 in PLoS Genetics the team shows that loss of ATRX is a common event in the genesis of ALT lines. They also show that ALT cell lines frequently undergo chromosomal changes and are impaired in their ability to detect and repair damage in their DNA.
“These hallmarks of ALT are expected to help the detection of ALT-type tumors in the clinic and may lead to ALT-specific treatments,” says Titia de Lange, Leon Hess Professor and head of the Laboratory of Cell Biology and Genetics at Rockefeller University, team leader and co-author of the paper. They may also help explain differential survival benefit of ALT tumors.”
“This work represents a remarkable collaborative effort among five laboratories at four different institutions,” says John Petrini, a member of Memorial Sloan-Kettering’s Molecular Biology Program, team member and co-author of the paper. “Each of us brought our specific expertise to bear on this intriguing and important problem.”
“It exemplifies the power of collaboration,” says William Hahn, a senior associate member of the Broad Institute, team member and co-author of the paper.
More information: www.starrcancer.org/