Improving the accuracy of cancer diagnoses: New spectroscopy technique could help doctors better identify breast tumors
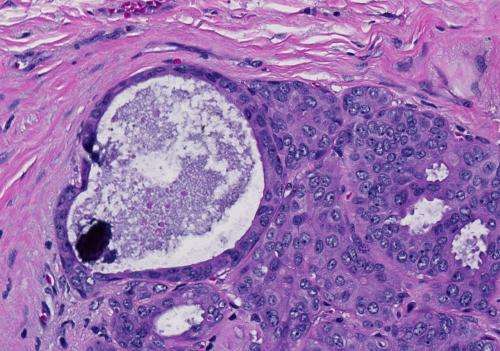
Tiny calcium deposits can be a telltale sign of breast cancer. However, in the majority of cases these microcalcifications signal a benign condition. A new diagnostic procedure developed at MIT and Case Western Reserve University (CWRU) could help doctors more accurately distinguish between cancerous and noncancerous cases.
When microcalcifications are spotted through mammography, doctors perform a follow-up biopsy to remove the suspicious tissue and test it for cancer. In 15 to 25 percent of cases, however, they are unable to retrieve the tissue that contains the calcium deposits, leading to an inconclusive diagnosis. The patient then has to undergo a much more invasive surgical procedure.
The new method, which uses a special type of spectroscopy to locate microcalcifications during the biopsy, could dramatically reduce the rate of inconclusive diagnosis, according to the researchers. In a study appearing in the Proceedings of the National Academy of Sciences the week of Dec. 24, they found that the spectroscopy technique had a success rate of 97 percent.
In addition, the spectroscopic approach could easily be integrated into the current biopsy procedure, says Ishan Barman, an MIT postdoc and one of the paper's lead authors. MIT postdocs Jaqueline Soares and Narahara Chari Dingari are also lead authors; senior authors are Maryann Fitzmaurice, senior research associate and adjunct associate professor of pathology and oncology at CWRU, and Ramachandra Rao Dasari, associate director of MIT's Laser Biomedical Research Center (LBRC).
'An arduous procedure'
Microcalcifications form when calcium from the bloodstream is deposited onto degraded proteins and lipids left behind by injured and dying cells. Though often seen in breast tumors, microcalcifications are rarely found in other types of cancer, Fitzmaurice says. Calcification also plays a major role in the hardening of the arteries seen in atherosclerosis.
Among women with microcalcifications spotted during a mammogram, only about 10 percent will turn out to have cancer, so the follow-up biopsy is critical. During that procedure, the radiologist first takes X-rays from three different angles to locate the microcalcifications, then inserts a needle into the tissue and removes five to 10 samples.
A pathologist then examines the tissues to see if they contain microcalcifications. If not, the radiologist tries again, after taking new X-rays. However, this second attempt is rarely successful, Fitzmaurice says.
"If they don't get them on the first pass, they usually don't get them at all," she says. "It can become a very long and arduous procedure for the patient, with a lot of extra X-ray exposure, and in the end they still don't get what they're after, in one out of five patients."
For the past several years, the MIT and CWRU team has been working to develop a spectroscopic technique that can analyze the tissue that the radiologist is about to biopsy—revealing, in a matter of seconds, whether that tissue actually contains microcalcifications.
They began with Raman spectroscopy, which uses light to measure energy shifts in molecular vibrations, revealing precise molecular structures. Because it offers such detailed information about the chemical composition of a tissue, Raman spectroscopy is very accurate in identifying microcalcifications. However, the equipment required is expensive, and the analysis takes a long time.
In the new study, the researchers showed that another technique, known as diffuse reflectance spectroscopy, gives results just as accurate as Raman spectroscopy. What makes diffuse reflectance spectroscopy more appealing is that it provides information within seconds, allowing the radiologist to move the needle if it's in the wrong spot, before taking any samples.
"With our new method, we could obtain similar results with less time and less expense," Dingari says.
Distinctive patterns
Diffuse reflectance spectroscopy works by sending light toward the tissue, then capturing and analyzing the light after its interaction with the sample. In this study, the researchers examined 203 tissue samples from 23 patients, within minutes of those samples' removal.
Each of the three types of tissue (healthy, lesions without microcalcifications, and lesions with microcalcifications) has subtle differences in its spectrographic signature, which can be used to distinguish among them. By analyzing these patterns, the researchers created a computer algorithm that can identify the tissues with a success rate of 97 percent.
The changes in tissues' light absorption are likely caused by altered levels of specific proteins (elastin, desmosine and isodesmosine) that are often cross-linked with calcium deposits in diseased tissue, Soares says.
For clinical use, a radiologist would perform spectroscopy just after inserting the needle to provide enhanced real-time guidance to the current biopsy procedure. The researchers are now planning for a study in which they will test their needle and spectroscopy setup in patients as the biopsies are being done.
James Tunnell, an associate professor of biomedical engineering at the University of Texas, says the findings represent a good first step toward creating a system that could have a big impact on breast cancer diagnosis. "This technology can be integrated into the system that is already used to take biopsies. It's a very simple technology that can get the same amount of accuracy as more complicated systems" such as Raman spectroscopy, says Tunnell, who was not involved in the study.
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.