New study sheds light on the complexity of gene therapy for congenital blindness
Independent clinical trials, including one conducted at the Scheie Eye Institute at the Perelman School of Medicine, have reported safety and efficacy for Leber congenital amaurosis (LCA), a congenital form of blindness caused by mutations in a gene (RPE65) required for recycling vitamin A in the retina. Inherited retinal degenerative diseases were previously considered untreatable and incurable. There were early improvements in vision observed in the trials, but a key question about the long-term efficacy of gene therapy for curing the retinal degeneration in LCA has remained unanswered. Now, new research from the Scheie Eye Institute, published this week in the Proceedings of the National Academy of Sciences, finds that gene therapy for LCA shows enduring improvement in vision but also advancing degeneration of affected retinal cells, both in LCA patients and animal models of the same condition.
LCA disease from RPE65 mutations has two-components: a biochemical blockade leading to impaired vision, and a progressive loss of the light-sensing photoreceptor cells throughout life of the affected patient. The authors of the new study explain that until now gene therapy has been optimistically assumed, but not proven, to solve both disease components at the same time.
"We all hoped that the gene injections cured both components – re-establishing the cycle of vision and also preventing further loss of cells to the second disease component" said Artur V. Cideciyan, PhD, lead author and co-investigator of an LCA clinical trial at Penn.
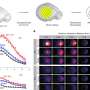
Yet, when the otherwise invisible cell layers of the retina were measured by optical imaging in clinical trial participants serially over many years, the rate of cell loss was the same in treated and untreated regions. "In other words, gene therapy improved vision but did not slow or halt the progression of cell loss," commented Cideciyan.
"These unexpected observations should help to advance the current treatment by making it better and longer lasting," commented co-author Samuel G. Jacobson, MD, PhD, principal investigator of the clinical trial. "Slowing cell loss in different retinal degenerations has been a major research direction long before the current gene therapy trials. Now, the two directions must converge to ensure the longevity of the beneficial visual effects in this form of LCA."
In a continuation of the longstanding collaboration between the Scheie investigators and the Section of Ophthalmology at Penn School of Veterinary Medicine headed by co-authors Gustavo D. Aguirre, VMD, PhD, and William A. Beltran, DVM, PhD, studies were performed to test whether the clinical results were also present in the canine model of this LCA at disease stages equivalent to those in human patients. "Our gene treatment in this canine model provided the groundwork for the clinical trials of patients, and now we added data to confirm the fact that retinal degeneration does continue despite improved vision" said Aguirre. "The next step is to perform the relevant experiments to ask what intervention will stop the degeneration if added to the gene therapy."
"These new findings contribute to greater clarity in understanding the natural history and complexity of the RPE65 form of LCA and provide a firm foundation for future investigations," said Joan M. O'Brien MD, professor and chair of the Department of Ophthalmology and director of the Scheie Eye Institute.