February 27, 2013 report
No genetic clock for neuron longevity
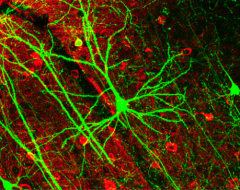
(Medical Xpress)—People are living longer than ever before, thanks to medical and technological advances. Unfortunately, aging can be associated with a decrease in brain function. This is because, unlike other cells in the body, neurons do not replicate. Neuroscientists in Italy have extended the lives of mouse neurons by injecting them into the brains of longer-lived rats, according to a study published in Proceedings of the National Academy of Sciences. This indicates that neuronal lifespan is not predetermined, but depends on conditions in the microenvironment.
After injecting green fluorescent protein into neuron precursor cells in the embryos of mice with an average lifespan of 18 months, Lorenzo Magrassi of the University of Pavia and his team transplanted the cells into the fetuses of Wistar rats, which live twice as long. Once the cells had differentiated, the researchers focused on a type of neuron called a Purkinje Cell (PC). Mice lose almost half of their PCs long before they reach old age. Wistar rats, on the other hand, keep almost all of their PCs until they die.
The researchers euthanized the rats after three years. When they examined the rats' brains, they found that the transplanted PCs had survived until the animals' deaths. The neurons showed the same signs of aging as neighboring rat neurons.
Although Magrassi and his colleagues were able to increase the lifespans of the transplanted neurons significantly, the transplantation did not alter all of the neurons' characteristics. The PCs retained the small size normally found in mice.
Magrassi says that the research proves that the longevity of neurons is not predetermined. Our brain cells do not have a genetic clock that tells them when to die. Instead, the microenvironment of the brain plays an important role in deciding how long neurons will live. He claims that by injecting neurons into species that live even longer, scientists could increase neuronal lifespan still more.
The research indicates that an aging body need not imply an aging brain. According to Magrassi, the findings suggest that neuronal implants could help people suffering from degenerative diseases such as Alzheimer's disease and Parkinson's disease.
Now, the team is trying to find out why PCs survive so much longer in rats than in mice by comparing the proteomes of the transplanted neurons with those of the host neurons. In an email to The Scientist, Gilbert Bernier, a molecular biologist at the University of Montreal who was not involved in the study, suggested that mice could be losing their PCs because of other types of neurons or because of inflammation caused by immune cells.
More information: Lifespan of neurons is uncoupled from organismal lifespan, PNAS, 2013. doi: 10.1073/pnas.1217505110
Abstract
Neurons in mammals do not undergo replicative aging, and, in absence of pathologic conditions, their lifespan is limited only by the maximum lifespan of the organism. Whether neuronal lifespan is determined by the strain-specific lifetime or can be extended beyond this limit is unknown. Here, we transplanted embryonic mouse cerebellar precursors into the developing brain of the longer-living Wistar rats. The donor cells integrated into the rat cerebellum developing into mature neurons while retaining mouse-specific morphometric traits. In their new environment, the grafted mouse neurons did not die at or before the maximum lifespan of their strain of origin but survived as long as 36 mo, doubling the average lifespan of the donor mice. Thus, the lifespan of neurons is not limited by the maximum lifespan of the donor organism, but continues when transplanted in a longer-living host.
(c) 2013 Medical Xpress

















