Study shows longer treatment for children with langerhans cell hystiocytosis improves survival rates
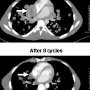
A new international study finds that prolonged, intense initial treatment in children with multi-system Langerhans cell histiocytosis (MS-LCH) can achieve survival rates as high as 84 percent—a full 15 percent improvement over the previous clinical trial in this series.
The study, LCH-III, is published in Blood, the journal of the American Society of Hematology. It is the third in a series of international randomized clinical trials for LCH that spans twenty years initiated and coordinated by the Histiocyte Society, a group of more than 200 physicians and scientists worldwide. The LCH clinical trial series are the first randomized clinical trials for the treatment of LCH.
Langerhans cells are found throughout the body and help regulate the immune system. In LCH, the cells proliferate excessively in a single organ or in multiple organ systems, damaging surrounding tissue and producing a wide range of symptoms. LCH occurs most often in children and its cause is unknown. In the most serious cases, LCH affects "risk organs," such as the liver or lungs, and can be fatal.
In this study, high-risk patients received one or two six-week courses of chemotherapy, followed by milder continuation therapy in those who responded to the initial course—for a combined 12 months of treatment. The overall five-year survival probability for these patients was 84 percent—substantially higher than in the two preceding trials (62 percent for LCH-I and 69 percent for LCH-II), in which patients were treated for six months. Moreover, the five-year risk of disease reactivation was much lower (27 percent) than in comparable patients in the two earlier trials.
Lower-risk patients in this study who responded after an initial six-week course of treatment were randomly assigned to receive six or 12 months' total treatment. The longer 12-month treatment significantly decreased the disease reactivation rate (37 percent), compared to the six-month treatment group (54 percent).
The study authors note that the first 12 weeks of treatment seem to be a critical time for patient outcomes with MS-LCH and that "aggressive salvage therapy was quite effective in patients not responding to the protocol therapy"—achieving a survival rate of 75 percent.
"These findings are real cause for hope for children with multi-system LCH, now that we have achieved improved survival rates in these three sequential studies," said senior author Stephan Ladisch, MD, a researcher in the Center for Cancer and Immunology Research at the Children's Research Institute and Professor of Pediatrics and Biochemistry/Molecular Biology at Children's National Medical Center. "The international collaboration in these trials has been essential to making significant progress against such a rare disorder, and children around the world will benefit from this research."
Results of the first LCH clinical trial were published in 2001 in the Journal of Pediatrics. Findings from the second trial were published in Blood in 2008. Goals for future trials include reduction of the remaining 15 percent mortality among patients with risk-organ involvement and reduction of the 30-40 percent disease reactivation rate.