MEND researchers are ready for human trials of an obesity drug showing dramatic results in mice
As sometimes happens in science, a medication used for one thing turns out to be very good for something else.
Such appears to be the case with the drug amlexanox, an asthma and canker sore medication, which has been shown by U-M researcher Dr. Alan Saltiel to inhibit two genes that play a role in metabolism and fat-burning in mice. And it may have the same effect on humans, potentially improving type 2 diabetes, insulin resistance, obesity, and non-alcoholic fatty liver disease (NAFLD).
Now the time has come to test the drug's effectiveness in humans. Elif Oral, M.D., Associate Professor of Internal Medicine in the Division of Metabolism, Endocrinology & Diabetes (MEND), is seeking people with type 2 diabetes or obesity and evidence of central obesity (belly fat) for this potentially ground-breaking study using amlexanox in humans. People can be on oral diabetes medication (no insulin) and should have a Body Mass Index (BMI) between 27 and 36 and an Hb A1c of less than 9.5%.
Since this is a "proof-of-concept" study in the early stages of this investigation, only 10 participants are needed. All test and drug costs are covered by the study and, in addition, the subjects will be compensated for their time.
If the drug is shown to work in humans, according to Dr. Oral, "This would mean that there is a completely different drug available for people that has a completely different mechanism of action in the body. If this drug can address weight and diabetes — as well as inflammation — without causing major side effects, this would be a huge innovation!"
Dr. Oral is also the director of the MEND Post-Bariatric Surgery Clinic and medical director of the U-M Health System's Bariatric Surgery Program and is an internationally recognized expert on lipodystrophy (a disorder of the fat cell hormone called leptin). Through her clinical practice at the MEND Clinic and her role in the Bariatric Surgery Program, she has seen first-hand the toll that obesity and diabetes take on people.
How did the idea to study amlexanox for such an off-label use come about? The lab of Dr. Alan Saltiel, Mary Sue Coleman Director of the U-M Life Sciences Institute (LSI), discovered that the genes IKKE and TBK1 play a crucial role for maintaining metabolic balance, first published in 2009 in the journal Cell.
Following this discovery, they searched for compounds that inhibit IKKE and TBK1, using high-throughput chemical screening at LSI's Center for Chemical Genomics. In this way, the researchers found the approved off-patent drug, amlexanox.
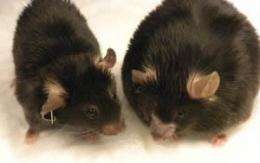
The Saltiel team then demonstrated that amlexanox had profound beneficial effects in both genetic and dietary-induced obese mice. The chemical lowered the weight of obese mice and reversed related metabolic problems such as diabetes and fatty liver. These findings were published this past February in the journal Nature Medicine.
Once the mouse findings were completed, Drs. Saltiel and Oral teamed up to translate these studies of amlexanox to humans in this clinical trial.
What's next? "After this first study," said Dr. Oral, "we will undertake a larger, simpler placebo-controlled trial and go after the effects that we will observe in the first study. I hope that this second study will start before the end of the year."
For the initial study, amlexanox is taken orally in a pill three times a day for a period of 12 weeks, followed by a follow-up four weeks after therapy ends. The researchers will evaluate the changes in metabolic parameters (e.g., blood cholesterol, liver function, insulin resistance) and body composition characteristics (e.g., the pattern of fat distribution in the body). Seven eligible subjects in this study will also be evaluated for a change in liver disease by undergoing a liver biopsy, if they so wish.
More information: "Clinical Protocol to Investigate the Efficacy of Amlexanox for Treatment of Glucose and Lipid Abnormalities in Obese Type 2 Diabetes"