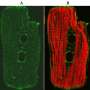
New research sheds light on abnormal heart muscle thickening and potential treatment
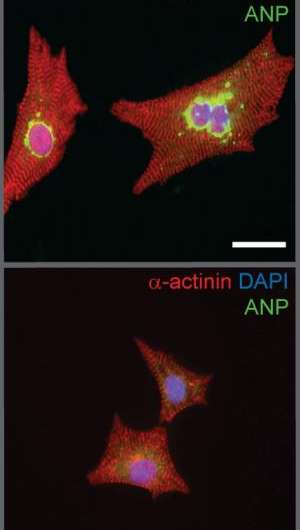
While most people would consider a big heart to be a good thing, for heart disease experts, it is often a sign of serious disease. Now, Dr. Lynn Megeney of the Ottawa Hospital Research Institute (OHRI) and the University of Ottawa (uOttawa) has made the surprising discovery that proteins involved in cell death also play a key role in abnormal heart muscle thickening. The research, published in the October 13, 2013 online edition of Proceedings of the National Academy of Sciences (PNAS), could lead to new treatments for certain forms of heart disease.
Heart muscle thickening (called cardiac hypertrophy) can be a healthy response to exercise and pregnancy; however, it often occurs in people with high blood pressure, diabetes, heart failure and certain genetic conditions. In these people, the heart can easily grow twice as large as normal, largely due to an increase in the size of individual heart muscle cells. While some muscle thickening can help the heart adapt (much like any muscle exposed to increased work), too much growth can lead to increased stiffness and reduced blood supply, and eventually reduced pumping function and heart failure.
Several years ago, Dr. Megeney noticed that heart muscle cells undergoing this kind of abnormal growth had many similarities with cells that are beginning to undergo an orderly form of cell suicide called programmed cell death. In the current research paper, Dr. Megeney and his team show that blocking the proteins that control this form of cell death also blocks abnormal heart muscle thickening.
Dr. Megeney and his team exposed rats to a number of different drugs that each induce abnormal heart muscle thickening. The rats were then given a form of experimental gene therapy to block cell suicide proteins in the heart. Three weeks later, the rats that received the experimental therapy had much smaller heart muscle cells (37 per cent smaller than those that did not receive the therapy), and smaller hearts overall. In fact, the disease model rats that received the experimental therapy seemed just as healthy as normal rats.
"Our research shows, for the first time, that heart muscle cells use the same molecular machinery for unhealthy growth as they would use to commit suicide," said Dr. Megeney, a senior scientist at OHRI and associate professor at uOttawa. "This may seem quite surprising to some people, but it fits with a growing body of research showing that cell death proteins can play many other roles in the body."
"Our research also shows, for the first time, that if we block the activity of cell suicide proteins in the heart, we can block abnormal heart muscle thickening in animal models," he added. "Although more research needs to be done, we think this may represent a promising new strategy for treating certain forms of heart disease. We are already investigating possible approaches to achieve this in humans, and we have identified some promising leads."
"This research is very important scientifically, and potentially clinically as well," said Dr. Duncan Stewart, a practicing cardiologist who is also CEO and scientific director of OHRI, vice-president of research at The Ottawa Hospital, professor at uOttawa. "This research is particularly applicable to certain genetic forms of heart disease, as well as to hypertension, which affects about 40 per cent of the adult population."
"An important observation from our work is that proteins from the caspase family, which play a key role in cell suicide, are also activated early in the process of cardiac cell hypertrophy," said Dr. Pasan Fernando, a co-author on the paper who is also a scientist at Nordion and the University of Ottawa Heart Institute and assistant professor at uOttawa. "By blocking one or several of these proteins, we may be able to not only reduce cardiac disease but also prevent it from even occurring."
More information: Intrinsic-mediated caspase activation is essential for cardiomyocyte hypertrophy. Charis Putinski, Mohammad Abdul-Ghani, Rebecca Stiles, Steve Brunette, Sarah A. Dick, Pasan Fernando, and Lynn A. Megeney. PNAS. Online early edition. October 13, 2013. www.pnas.org/cgi/doi/10.1073/pnas.1315587110