New clues to memory formation may help better treat dementia

Do fruit flies hold the key to treating dementia? Researchers at the University of Houston (UH) have taken a significant step forward in unraveling the mechanisms of Pavlovian conditioning. Their work will help them understand how memories form and, ultimately, provide better treatments to improve memory in all ages.
Gregg Roman, an associate professor of biology and biochemistry at UH, and Shixing Zhang, his postdoctoral associate, describe their findings in a paper titled "Presynaptic Inhibition of Gamma Lobe Neurons Is Required for Olfactory Learning in Drosophila," appearing Nov. 27 in Current Biology, a scientific bimonthly journal published by Cell Press.
"Memory is essential to our daily function and is also central to our sense of self," Roman said. "To a large degree, we are the sum of our experiences. When memories can no longer be retrieved or we have difficulty in forming new memories, the effects are frequently tragic. In the future, our work will enable us to have a better understanding of how human memories form."
Roman and Zhang set about to unravel some of these mysteries by studying the brains of fruit flies (Drosophila). Within the fly brain, Roman says, there are nerve cells that play a role in olfactory learning and memory. Olfactory learning, he says, is an example of classical conditioning first described by Pavlov in his experiment with dogs. In their study, the flies were trained to associate a weak electric shock with an odor. After training, the flies avoided that odor.
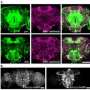
"We found that these particular nerve cells – the gamma lobe neurons of the mushroom bodies in the insect brain – are activated by odors. Training the flies to associate an odor with an electric shock changed how these cells responded to odors by developing a modification in gamma lobe neuron activity, known as a memory trace," he said. "Interestingly, we found that training caused the gamma lobe neurons to be more weakly activated by odors that were not paired with an electric shock, while the odors paired with electric shock maintained a strong activation of these neurons. Thus, the gamma lobe neurons responded more strongly to the trained odor than to the untrained odor."
The team also showed that a specific protein – the heterotrimeric G(o) protein – is naturally involved in inhibiting gamma lobe neurons. Roman says removing the activity of this protein only within the gamma lobe neurons resulted in a loss of the memory trace and, thus, poor learning. Therefore, inhibiting the release of neurotransmitters from these neurons through the actions of the G(o) protein is key to forming the memory trace and associative memories.
The significance of using fruit flies is that while their brain structure is much simpler with far fewer neurons, the mushroom body is analogous to the perirhinal cortex in humans, which serves the same function of sensory integration and learning. This simplicity allows scientists to gain insights into how memories are acquired, stored and retrieved.
"Drosophila represents the Goldilocks principle of neural research, with sufficient behavioral complexity, while maintaining a huge advantage in neural simplicity," Roman said. "The complex behaviors allow us to examine many behavioral processes like learning, attention, aggression and addiction-like behaviors, while the simplicity allows us to dissect the crucial neural activities down to single cells. Additionally, Drosophila has the most powerful genetic toolkit available for behavioral experimentation. In using these tools, we are genetically identifying the molecules necessary to perform these behaviors and dissecting the logic of the neural circuits that allow for changes in behavior to occur."
The pair says all their experience to date suggests the molecules and logic will translate to most animals, including humans, leading to a more complete understanding of how memories form in humans, both at the level of molecules and through the activity of neural circuits.
Initially funded by a Norman Hackerman Advanced Research Program award from the state of Texas, a Major Research Instrumentation Program grant from the National Science Foundation was also critical, supplying the microscope necessary for measuring neural activity.