Scientists create potential vaccine against respiratory syncytial virus

Scientists at The Scripps Research Institute (TSRI) have invented a new method for designing artificial proteins, and have used it to make key ingredients for a candidate vaccine against a dangerous virus, respiratory syncytial virus (RSV), a significant cause of infant mortality. The virus has been resistant to current vaccine-design strategies.
With the help of collaborating laboratories, the scientists were able to apply the new method, which uses a "rational design" approach to making vaccines focused on specific binding areas (epitopes) on the virus. The result was designer vaccine proteins that the scientists showed stimulate the production of the desired virus-neutralizing antibodies in rhesus macaques.
"This was a proof-of-principle demonstration of a technology that could be very useful against HIV, influenza and other highly variable viruses that have been difficult to stop using traditional vaccine-design strategies," said William R. Schief, associate professor of immunology at TSRI.
The research is reported in by the journal Nature on February 5, 2014.
Folding from Loops
The new protein-design method represents a significant advance over previous methods.
"One approach we and others have taken has been to transplant a protein fragment of interest, for example one that mimics a particular structure on a virus, onto an existing protein 'scaffold,'" said TSRI Research Associate Bruno E. Correia, a member of the Schief laboratory at the time of the study and lead author of the new report. "While this approach often works well to mimic the structure of a viral epitope, it has never successfully induced neutralizing antibodies, and in some cases this method falls short of even producing viable vaccine candidates."
In these difficult cases, the scaffold structure fails to stabilize the transplanted fragment, resulting in an imperfect mimic of the virus and consequent loss of immune-stimulating properties.
The TSRI scientists wanted a way to design scaffold proteins from scratch—proteins that would fit around their functional fragments more naturally, and would do a better job of stabilizing them.
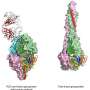
The result was a new software app, "Fold from Loops," for designing proteins that fold up around a functional fragment of interest. For a proof-of-principle demonstration, the scientists decided to attempt one of the most important current protein-design challenges: making a new protein that mimics a particular epitope on a virus, and thus can serve as a key component of a vaccine.
The Promise of Rational Vaccine Design
Researchers want to be able to stimulate antibody reactions against highly specific epitopes because some infectious agents seem unstoppable by traditional methods of immunization.
"The achievement announced today represents the confluence of recent technological advances in computational biology, structural biology and immune monitoring, and offers great potential for accelerating development of next generation vaccines against major global diseases," said Wayne C. Koff, chief scientific officer at IAVI, the International AIDS Vaccine Initiative, which helped to fund the studies.
Virtually all existing viral vaccines, for example, use whole (killed or weakened) virus particles or entire viral proteins to stimulate antibody reactions. These vaccines display virtually the same large set of viral epitopes that the immune system would encounter during a natural infection.
Yet some viruses, such as HIV and influenza viruses, effectively conceal their truly vulnerable epitopes during natural infections, displaying mostly "decoy" structures that change from one viral generation to the next. Only vaccines that can artificially stimulate large numbers of antibodies—against just the vulnerable epitopes—are likely to provide broad protection against such viruses.
Scientists know how to sift through blood samples of virus-exposed patients to find the rare, "broadly neutralizing" antibodies that hit those vulnerable epitopes. They also know how to map the precise atomic structures of these antibodies and their corresponding epitopes, using X-ray crystallography.
"What we haven't been able to do is to take that information about broadly neutralizing antibodies and their epitopes and translate it into effective, epitope-focused vaccines," said Correia.
A Candidate RSV Vaccine
For the test case, the team used the Fold from Loops software to design proteins that incorporate and stabilize a broadly neutralizing epitope on respiratory syncytial virus (RSV), a significant cause of infant mortality for which no preventive vaccine is yet available. Winnowing thousands of design possibilities down to four that seemed to have the best properties, the team turned them over to collaborating laboratories for preclinical testing and analysis.
In rhesus macaque monkeys, whose immune systems are quite similar to humans', the designer "immunogen" proteins showed great promise. After five immunizations, 12 of 16 monkeys were producing robust amounts of antibodies that could neutralize RSV in the lab dish.
"It's unusual to take a newly designed protein and immunize rhesus macaques with it," said Schief. "We were fortunate to collaborate with Philip Johnson at Children's Hospital in Philadelphia, whose laboratory performed those immunizations."
Analyses of the animals' immune responses were conducted at Johnson's laboratory and at the laboratory of James E. Crowe, Jr., at Vanderbilt University Medical Center and in Barney Graham's lab at the NIH/NIAID Vaccine Research Center.
At the laboratory of Roland K. Strong at Fred Hutchinson Cancer Research Center in Seattle, researchers performed X-ray crystallography of two neutralizing monoclonal antibodies produced by the macaques—antibodies that had never been described before—and confirmed that each hit the desired virus epitope.
Having proven the principle of epitope-specific design, Schief and his colleagues now hope to continue this line of research and produce a working RSV vaccine. "RSV is estimated to cause nearly seven percent of all human deaths worldwide in children ages one month to one year," said Schief. "Beyond that, RSV sends millions of kids to the hospital and right now there is no licensed vaccine. So we are going to push hard to see if we can make a vaccine for infants and children using these new technologies. We're also trying to improve this protein design method further and apply it to other vaccine projects including HIV and influenza vaccines."
More information: Proof of principle for epitope-focused vaccine design, Nature, DOI: 10.1038/nature12966