April 14, 2014 weblog
Transplanting interneurons: Getting the right mix
(Medical Xpress)—Despite early optimistic studies, the promise of curing neurological conditions using transplants remains unfulfilled. While researchers have exhaustively cataloged different types of cells in the brain, and also the largely biochemical issues underlying common diseases, neural repair shops are still a ways off. Fortunately, significant progress is being made towards identifying the broader operant principles that might bear on any one disease work-around. A review just published in Science focuses on recent work on transplanting interneurons—a diverse family of cells united by their mutual love of inhibition and their local loyalty. The UCLA-based authors, reach the conclusion that the fate of transplanted neurons ultimately depends less on the influences of the new host environment, and more on the early upbringing of the cells within the donor embryo.
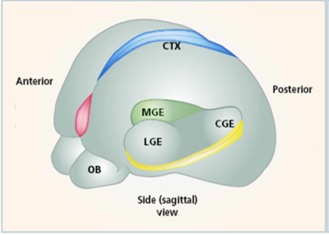
Interneurons are born in the lateral (LGE) and medial ganglionic eminence (MGE). Those that eventually colonize the cortex need to migrate a fairly long distance tangentially to get there, but once they arrive, they prefer to extend only local connections. By comparison, the excitatory pyramidal cells which end up sending long-range projections, are born within the cortex itself. Researchers have found that only those interneurons from the MGE have what it takes to make long migratory journeys. LGE neurons, when transplanted into postnatal host brains, remain in tight clumps whereas those from the MGE disperse throughout the cortex.
More importantly, it is now appreciated that transplanted interneurons closely follow cell-intrinsic programs rather than relying on host-specific cues to govern their survival and differentiation. The once popular conception of a life and death competition for neurotrophic factors, if at play here at all, seems to be a minor influence. Herds of transplanted neurons are still thinned in the host, for example, but those that die off do so asynchronously from the endogenous interneurons, and in line with their own internal programming. Scrutinous cell accounting has shown that after transplantation, the total number of interneurons within the host tissue greatly exceeds the nominal amount normally found.
An excess balance of inhibitory cells has been seen as desireable from the point of view of treating mismatches in excitability of the kind found in diseases like epilepsy. It is important to realize however, that binary electrical tallies only represent one aspect of neuronal function. Furthermore, in epilepsy, we might more generally view exciteability as just the readily observeable tip of underlying metabolic imbalance. None the less, suppression of spontaneous seizures in a mouse channelopathy model (mutant for a potassium channel known as Kv1.1) has been acheived with interneuron tranplants. In yet another case of nomenclature gone wild, this particular mutant has been associated with human interneuronopathies leading to severe tonic-clonic seizures.
Synapse constitution—number, type or strength of synapses—can be tough to quantify objectively and exactly. There have been indications that transplanted interneurons make 3X the number of synapses as native interneurons, but they are only one-third as strong as would be normally expected. The keyword here is "strong." There can be any combination of synaptic capabilities involved in this idea, things like electrical amplititude, reliability, or persistence at a high rate of firing all come into play in the idea of strength. The Chandelier cells control the axon initial segments well known and idiosyncratic interneuron known as the Chandelier cell, for example, commands access to the highly coveted axon initial segment where it effectively exercises complete veto over its associated pyramidal cell.
To increase the efficiency and fidelity of harvesting exact precursor cell subtypes, techniques like fluorescence-activated cell sorting (FACS) have been used in sample processing. Flourescent proteins under the control of forebrain or MGE specific promotors can be used to select individual cells types for later transplantation. To bias cells into somatostatin or parvalbumin-expressing populations, for example, wild-type MGE cells can then be exposed to sonic hedgehog or other fate-ruling factors.
Transplanting different kinds of cells together will probably be necessary to properly treat many diseases. Even non-neural cells like astrocytes and microglia may be critical to have in the mix. Exciting results obtained in mice last year, indicate that these cell types can thrive not just when transplanted across individuals but across species. The goal for the present time is to define good protocols for integrating one cell type first. Nimble cells that migrate well within the host, yet confine their influence to the local environment might be the most sensible place to start.
More information: Interneurons from Embryonic Development to Cell-Based Therapy, Science 11 April 2014: Vol. 344 no. 6180. DOI: 10.1126/science.1240622
© 2014 Medical Xpress