New headway in battle against neurodegenerative diseases
(Medical Xpress)—Conditions which may accelerate the spread of Parkinson's disease, and a potential means of enhancing naturally-occurring defences against neurodegenerative disorders, have been identified in two new studies.
Two significant breakthroughs which could inform future treatments for neurodegenerative diseases such as Alzheimer's and Parkinson's, have been announced by scientists.
The research, published in two separate studies this week, advances understanding of the early development of such disorders and how they might be prevented – in particular by identifying the biological areas and processes that could be pinpointed by future drugs.
Both sets of results have emerged from collaborations between the research groups led by Chris Dobson, Tuomas Knowles and Michele Vendruscolo at the University of Cambridge, who focus on understanding protein "misfolding" diseases. These include Alzheimer's and Parkinson's diseases, as well as numerous others.
The first study provides evidence that the early spread of the protein aggregates associated with Parkinson's appears to happen at an accelerated rate in mildly acidic conditions. This suggests that particular compartments within brain cells, which are slightly more acidic than others, may turn out to be appropriate targets for future treatments fighting the disease.
Meanwhile, researchers behind the second study appear to have identified a way in which the effectiveness of so-called molecular "chaperones", responsible for limiting the damage caused by misfolded proteins, can be significantly enhanced.
The papers appear in the latest issue of Proceedings of the National Academy of Sciences.
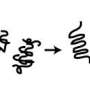
As the term suggests, protein misfolding diseases stem from the fact that proteins, which need to fold into a particular shape to carry out their assigned function in the body, can sometimes misfold. In certain cases these misfolded proteins then clump together into fibre-like threads, called amyloid fibrils, potentially becoming toxic to other cells.
How this formation begins at a molecular level is still not completely understood, but comprehending the process will be fundamental to the development of future therapies and is the subject of extensive current research.
The first of the new studies builds on research published in 2013, which showed that in Alzheimer's sufferers, the initial "nucleation" between proteins, which leads to amyloid formation, is followed by an amplification process called secondary nucleation. In these secondary events, the existing amyloid structures facilitate the formation of new aggregates, leading to their exponential increase. This process is likely to be at the heart of the development and spread of the disease in affected brains.
Using the same techniques, the researchers behind the latest study identified a similar process that is relevant in the early stage development of Parkinson's Disease. Their work focused on a protein called α-synuclein, which is associated with the disorder, and simulated different conditions in which this protein might misfold and form clumps.
As with the previous study on Alzheimer's, the research identified that Parkinson's could spread through a series of secondary nucleation events. In addition, however, it showed that in the case of α-synuclein, this happens at a highly accelerated rate only in solutions which are mildly acidic, with a pH below 5.8. The finding is important because certain sub-compartments within cells are more acidic than others, meaning that these may be particularly productive areas for future treatments to target.
Dr Tuomas Knowles, from the Department of Chemistry and a Fellow of St John's College, Cambridge, said: "This tells us much more about the molecular mechanisms underlying protein aggregation in Parkinson's and suggests that mildly acidic microenvironments within cells may enhance that process by several orders of magnitude. Not every sub-cellular compartment offers these conditions, so it takes us much closer to understanding how the disease might spread."
The second study meanwhile suggests a potential route to improving the effectiveness of a particular molecular "chaperone" – a loose classification for proteins which assist in the folding of others, thereby preventing them from causing damage when they misfold.
The researchers focused on a chaperone called α2-macroglobulin (α2M), which is found outside cells themselves. This is important because neurodegenerative diseases often stem from a process which begins with extracellular misfolding. The α2M was tested on a substrate of the amyloid-beta peptide associated with Alzheimer's Disease.
Typically, the potency of α2M is limited. The new study, however, found that when it comes into contact with the oxidant hypochlorite – the same chemical found in household bleach, which also naturally occurs in our immune systems – its structure is modified in a manner that makes it into a much more dynamic defence.
In their report, the researchers suggest that this increased effectiveness stems from the fact that α2M, which is usually found in a four-part, "tetrameric" form, breaks down into "dimeric", two-part forms when it comes into contact with hypochlorite.
The chaperone usually plays its role by preventing a misfolded protein from interacting with the membranes that surround and protect cells. Once in its dimeric form, however, receptor binding sites within the α2M are exposed, leading to specific interactions with receptors on the cell itself. If the α2M has already interacted with misfolded proteins, this connection triggers the cell to break the potentially harmful protein down.
"It's almost like a warning flag for the cell, telling it that something is wrong," Dr Janet Kumita, from the Department of Chemistry, explained. "It triggers the cell to react in a way that subjects the cargo of misfolded protein to a degradation pathway."
"Increasing its potency in this way is an exciting prospect. If we could find a way of developing a drug that introduces the same structural alterations, we would have a therapeutic intervention capable of increasing this protective activity in patients with Alzheimer's Disease."
Professor Christopher Dobson, from the University's Department of Chemistry and Master of St John's College, said: "These studies add very substantially to our detailed understanding of the molecular origins of neurodegenerative diseases, which are now becoming one of the greatest threats to healthcare in the modern world."
"We are beginning to understand exactly how a single, aberrant event can lead to the proliferation and spreading of toxic species throughout the brain, and the manner in which our sophisticated defence mechanisms do their best to suppress such phenomena. It will undoubtedly provide vital clues to the development in due course of new and effective drugs to combat these debilitating and increasingly common disorders."