Researchers discover key factor in early auditory system development
Researchers at the National Institutes of Health have uncovered a molecule in an animal model that acts as a key player in establishing the organization of the auditory system. The molecule, a protein known as Bmp7, is produced during embryonic development and acts to help sensory cells find their ultimate position on the tonotopic map, which is the fundamental principle of organization in the auditory system. The tonotopic map groups sensory cells by the sound frequencies that stimulate them. The study is the first to identify one of the molecular mechanisms that determines position.
Findings from the study, led by Zoe F. Mann, Ph.D and Matthew W. Kelley, Ph.D., of the Laboratory of Cochlear Development at the National Institute on Deafness and Other Communication Disorders (NIDCD), were published in the May 20, 2014 issue of Nature Communications. The research was performed in collaboration with scientists from the University of Virginia (UVa) School of Medicine, Charlottesville, and Imperial College in London. The American Hearing Research Foundation provided additional support.
An additional study, appearing in the same edition, is led by NIDCD-supported researchers Benjamin R. Thiede, Ph.D., and Jeffrey T. Corwin, Ph.D. at UVa. Working in collaboration with Drs. Mann and Kelley, the researchers reveal that another signaling molecule, retinoic acid, acts in concert with Bmp7 to position cells.
"The findings could open doors to therapies that take advantage of Bmp7's navigational talents to direct the formation of regenerated sensory cells that are tuned to respond to a specific frequency," says James F. Battey, Jr., M.D., Ph.D., director of NIDCD. "Since many forms of hearing loss are limited to specific frequencies, this approach could lead to replacement sensory cells that are tailored to individual needs."
The human ear can detect a wide range of frequencies, from the low rumble of distant thunder to the high-pitched whine of a mosquito. The sensory cells that detect these sounds are called hair cells, named for the hair-like strands that cluster on their tops. Hair cells are spread across a flat surface called the basilar membrane, which rolls up like a carpet and tucks into a snail shell-shaped structure in the inner ear called the cochlea.
Part of what accounts for our remarkable range of hearing is that hair cells have different specializations. Rather than working to sense all audible frequencies, each of our roughly 16,000 hair cells is dedicated to a narrow range. Hair cells are ordered along the basilar membrane's length, or axis, according to the frequency they detect. Those that sense low pitches are at one end and those that detect high-frequency sounds are at the opposite end. The cells in between step through the mid-range pitches.
This spatial arrangement of hair cells on the basilar membrane–the tonotopic map—has been known for years. What hasn't been known is how each hair cell learns to "hear" specific frequencies.
"During development, hair cells at each position along the axis need to figure out where they are so that they know what frequency they should be listening to," said Dr. Kelley. "This is called positional identity. We wanted to know how hair cells figure out their position."
Dr. Kelley suspected that, like numbers on a ruler, the positions of hair cells along the basilar membrane were marked by stepwise differences in the level of a signaling molecule that would determine position. Molecular concentration gradients of this sort have been shown to steer the positioning of other cell types in the body during development.
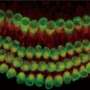
To see if such a signaling molecule might be involved in the structural organization of the cochlea, Dr. Mann examined the basilar papilla from six-day old chick embryos. The basilar papilla in chickens is similar to the cochlea in mammals, with hair cells arranged along the length of its basilar membrane in a similar fashion according to frequency. The researchers reasoned that if a molecular concentration gradient were involved in positioning hair cells, the molecule's level would be higher at one end of the basilar papilla than the other.
When they split the basilar papilla in half looking for molecules, one stood out because of the striking difference in its level between the two halves—Bmp7—a signaling protein known to play a role in the development of bone and kidneys. Additional experiments revealed a gradual gradient in the level of Bmp7 across the length of the basilar papilla.
The researchers next showed that Bmp7 promotes the development of low-frequency-sensing hair cells. When they bathed developing basilar papillas in a solution containing Bmp7, they found that all the hair cells developed characteristics of low-frequency-sensing hair cells, even those at the high-frequency end.
These findings suggest that during embryonic development, high levels of Bmp7 at one end of the basilar papilla signal the formation of low-frequency-sensing hair cells. Decreasing levels of Bmp7 along the length of the basilar papilla map with a gradual change towards tuning to higher frequencies.
In future work, Dr. Kelley and his team aim to use a mouse model to understand the role of Bmp7 in specifying the positioning of hair cells in a mammalian organism. Bmp7 is known to be present in cells of the inner ear in mammals, suggesting a possible role for the molecule in tuning. The researchers hope to be able to outline its precise role in patterning parts of the auditory system.
"The entire auditory system is assembled according to individual frequencies," said Dr. Kelley. "Complex sounds like music or speech that consist of many different frequencies are split into individual frequencies in the ear, processed through separate channels, and then reassembled in the brain. By revealing the part played by Bmp7 in patterning hair cells in the inner ear, we may have uncovered a broader role for the molecule in the auditory system as a whole."