Researchers identify new protein linked to leukaemia growth

(Medical Xpress)—Their work has identified a protein called PIP4K2A that could be a new target in drug development.
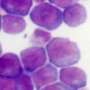
Uncontrolled growth of cells is one of the hallmarks of cancer. In acute myeloid leukaemia, abnormal white blood cells accumulate in the bone marrow, blocking production of normal red and white blood cells.
All blood cells are derived from so-called haematopoietic stem cells, but there are certain molecules – phosphoinositides – that appear to control potential conversion of these stem cells into cancerous leukaemic cells. Some of these phosphoinositides switch on specific cell signalling pathways, resulting in rapid growth and enhanced survival.
Regulation of these phosphoinositides (PIs) is carried out by a variety of proteins – known as PI modulators. Now researchers based at the Cancer Research UK Manchester Institute at The University of Manchester – part of the Manchester Cancer Research Centre – have explored the role of a variety of these genes to see which PI modulators have a key role in leukaemia.
Dr Tim Somervaille, who led the research, said: "Little is known about the role of PI modulators in leukaemia. We wanted to find out which ones were responsible for cell growth or survival in acute myeloid leukaemia."
The group looked at human acute myeloid leukaemia cells and switched off individual genes in a process known as targeted knockdown.
They found that one PI modulator, a protein known as PIP4K2A, was essential for the growth of leukaemia cells – both those grown in the lab and those from patient samples. When this protein was switched off, the leukaemia cells died.
"Developing new treatments for cancer is based on a better understanding of what makes cancer cells grow and thrive so that we can deprive them of something essential for their survival. Importantly, in normal cells, knockdown of the PIP4K2A protein had no adverse effect – this makes it an ideal target for future drug development in leukaemia," added Dr Somervaille.