Signal from fat tissues improves insulin secretion in diabetic mice, suggests potential therapy
Researchers have discovered that adipsin, a cell signaling protein made by fat cells, plays a critical, previously unsuspected role in stimulating insulin secretion to control blood sugar, according to a report from Dana-Farber Cancer Institute and Karolinska Institutet.
Bruce Spiegelman, PhD, senior author of the report in Cell, said the discovery could have implications for treatment of type 2 diabetes, a growing epidemic affecting an estimated 382 million people around the world.
Per-Olof Berggren, PhD, and professor at Karolinska Institutet in Stockholm, Sweden, and a co-investigator of the study, added that adipsin "might be the long-sought molecule linking fat tissue metabolism to pancreatic beta cell function."
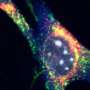
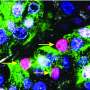
In an experiment with obese, diabetic mice that lacked adipsin, replacing the protein improved the health of beta cells in the pancreas, which normally secrete insulin but malfunction in severe diabetes.
The mice had been genetically altered to lack adipsin, but the scientists also discovered that adipsin is deficient in human patients with severe type 2 diabetes.
"This suggests a new approach to treating type 2 diabetes in patients whose pancreatic beta cells work poorly, leaving them dependent on injected insulin," said Spiegelman, of Dana-Farber's Cancer Biology Department and a professor at Harvard Medical School.
"If humans respond similarly to the mice in this study," he said, "correcting their deficiency of adipsin would improve beta cell function and perhaps maintain enough natural insulin production to avoid or delay having to take additional insulin."
Checking for adipsin levels in diabetic patients might help doctors predict which individuals are at highest risk of impending beta cell failure so that treatment could be started earlier, the authors noted.
James Lo, MD, PhD, a cardiologist in the Spiegelman lab, is the report's first author. In addition to the Karolinska Institutet, other authors of the report are from the University of Leipzig, Germany, and the University of Ancona, Italy.
Adipsin, the first member discovered of a class of proteins called adipokines, which are secreted into the bloodstream by adipocytes, or fat cells, and continuously circulate to influence a variety of metabolic and immune functions. Adipsin was discovered by Spiegelman's research group in 1987, and was found to be a component of the immune system, but its newly identified role in controlling insulin production by the pancreas was entirely unsuspected.
The Spiegelman lab and other researchers have recently found that some immune components are present in fat cells and are involved in metabolic energy balance. This growing area of investigation prompted Lo and Spiegelman to revisit the function of adipsin. Lower levels of adipsin had been reported in obese and diabetic animals, but unchanged or elevated levels in overweight and diabetic humans, leaving unclear how the protein functions in those conditions.
In this study, the Dana-Farber investigators used "knockout" mice lacking the adipsin gene and "wild type" mice with normal adipsin levels. Both types of animals became obese on a high-fat diet and developed excess blood sugar – a pre-diabetic state. The symptoms were worse in the adipsin knockout mice than the wild-type animals, which had normal adipsin activity.
In the report the scientists noted that the difference in symptoms is explained by "an unexpected and striking requirement of adipsin for proper insulin secretion by the pancreatic beta cells."
Since adipsin is secreted into the circulation by fat cells, said Lo, "it makes sense that when an individual's fat mass increases in obesity, that would increase secretion of adipsin to help compensate" by stimulating beta cells to produce more insulin.
Manipulation of this molecular switch may serve as a novel therapy in type 2 diabetes, the researchers wrote.
One of adipsin's effects is to generate a peptide called C3a that flows through the blood stream and binds to a C3 receptor on pancreatic beta cells, triggering secretion of insulin. Berggren noted, "This is particularly interesting since it clearly points to an important cross talk between peripheral tissues like the fat tissue and the insulin secreting beta cell in the regulation of glucose homeostasis."
More information: "Adipsin is an Adipokine that Improves β Cell Function in Diabetes" James C. Lo, Sanda Ljubicic, Barbara Leibiger, Matthias Kern, Ingo B. Leibiger, Tilo Moede, Molly E. Kelly, Diti Chatterjee Bhowmick, Incoronata Murano, Paul Cohen, Alexander S. Banks, Melin J. Khandekar, Arne Dietrich, Jeffrey S. Flier, Saverio Cinti, Matthias Blüher, Nika N. Danial, Per-Olof Berggren, Bruce M. Spiegelman. Cell Volume 158, Issue 1, 3 July 2014, DOI: 10.1016/j.cell.2014.06.005