Study links enzyme to Alzheimer's disease
Unclogging the body's protein disposal system may improve memory in patients with Alzheimer's disease (AD), according to a study from scientists at Kyungpook National University in Korea published in The Journal of Experimental Medicine.
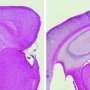
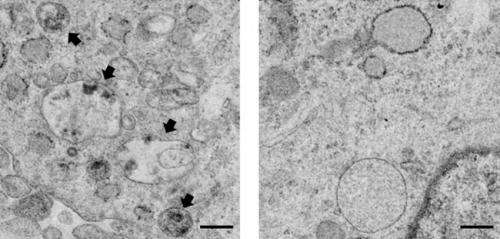
In AD, various biochemical functions of brain cells go awry, leading to progressive neuronal damage and eventual memory loss. One example is the cellular disposal system, called autophagy, which is disrupted in patients with AD, causing the accumulation of toxic protein plaques characteristic of the disease. Jae-sung Bae and colleagues had earlier noted that the brains of AD patients have elevated levels of an enzyme called acid sphingomyelinase (ASM), which breaks down cell membrane lipids prevalent in the myelin sheath that coats nerve endings. But whether increased ASM directly contributes to AD (and if so, how) was unclear.
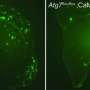
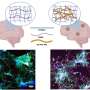
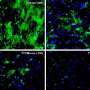
The group now finds that these two defects are linked. In mice with AD-like disease, elevated ASM activity clogged up the autophagy machinery resulting in the accumulation of undigested cellular waste. Reducing levels of ASM restored autophagy, lessened brain pathology, and improved learning and memory in the mice. Provided these results hold true in humans, interfering with ASM activity might prove to be an effective way to slow—and possibly reverse—neurodegeneration in patients with AD.
More information: Lee, J.K., et al. 2014. J. Exp. Med. DOI: 10.1084/jem.20132451