Researchers pinpoint protein hub necessary for proper brain development
Researchers at the UNC School of Medicine have found that the protein glycogen synthase kinase-3, or GSK-3, is crucial for proper brain development early in life, a surprising finding considering that drug companies are searching for ways to limit the protein's function to treat conditions such as bipolar disorder, schizophrenia, and Alzheimer's disease.
The discovery, published today in the journal eLife, also has implications for the study of neurodevelopmental disorders, which can result from abnormal brain formation during embryonic stages and infancy.
"We found that deleting GSK-3 from a specific type of neuron disrupts how those neurons migrate through the cortex," said Meghan Morgan-Smith, PhD, a recent UNC graduate and first author of the eLife paper. Neuron migration is key to how the cerebral cortex becomes organized so it can function properly in adulthood. "The finding was shocking. We thought that deleting GSK-3 would enhance the migration of the neurons. Instead, the opposite happened."
Until now, scientists have viewed GSK-3 as an important drug target because it regulates the function of about 200 other proteins in different kinds of brain cells. In certain conditions, such as schizophrenia and Alzheimer's disease, GSK-3 is thought to inhibit these proteins to a greater degree than is normal. By targeting GSK-3, pharmacologists try to suppress GSK-3's activity so that some of the downstream proteins are released and provide therapeutic effects. This is how, for instance, scientists think lithium works on GSK-3 to treat bipolar disorder.
William Snider, MD, director of the UNC Neuroscience Center and senior author of the eLife paper, said, "Our study shows that we need to be careful when inhibiting GSK-3 because the brain requires its activity for certain developmental and physiological processes."
Snider and Morgan-Smith said that several other proteins had been previously implicated in brain layer formation, but GSK-3 could wind up being the most important regulator of the entire process because it's a hub of cell signaling for so many other proteins.
"We think this finding is the tip of the iceberg in terms of what GSK-3 does to neurons," Snider said. "It likely affects the regulation of where axons extend and what synapses do to brain plasticity, which are critical for normal brain development."
In the embryonic human brain, neurons are born from progenitor cells and then migrate to the outer reaches of the cortex. As new neurons are born, they migrate past the previously-born neurons to their final position. The last neurons born – cells called layer 2/3 excitatory pyramidal neurons – migrate past all other neurons to form a new layer just under the skull. In this fashion, neurons form cortical layers that are responsible for higher brain functions, such as learning and memory.
During this layering process, the neurons form axons – long, arm-like extensions that connect to other cells in the cortex and elsewhere in the brain. When cortical layering goes awry, these axon connections don't form properly. The result can lead to a range of neurodevelopmental disorders, such as intellectual disabilities.
This same layering process occurs in mice, which also have layer 2/3 excitatory pyramidal neurons with GSK-3.
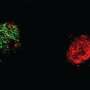
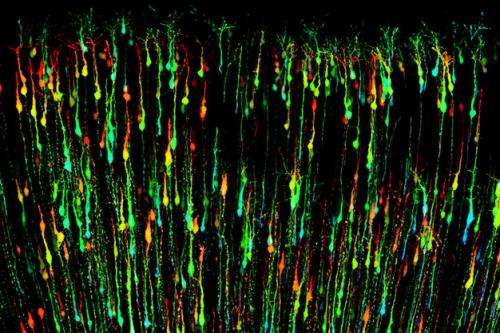
Using mouse genetics and a technique called in utero electroporation, Morgan-Smith was able to delete the GSK-3 gene from just the layer 2/3 excitatory pyramidal neurons and then study what happens to these cells during brain development.
"Deleting GSK-3 completely shut down the system," said Morgan-Smith, who conducted the research while a graduate student in Snider's lab. "The outer cortical layers aren't formed at all. The neurons are scattered throughout the cortex."
Morgan-Smith's research is the first to study the role of GSK-3 in layer 2/3 excitatory pyramidal neurons in a living system. Other studies of GSK-3 were done in cell cultures. Her work took 3 ½ years, and the published work is her dissertation. She's now finishing work in Snider's lab to figure out if deleting GSK-3 affects the way neurons signal to each other.
More information: eLife 2014;3:e02663. DOI: 10.7554/eLife.02663