Early antibiotic exposure leads to lifelong metabolic disturbances in mice
A new study published today in Cell suggests that antibiotic exposure during a critical window of early development disrupts the bacterial landscape of the gut, home to trillions of diverse microbes, and permanently reprograms the body's metabolism, setting up a predisposition to obesity. Moreover, the study shows that it is altered gut bacteria, rather than the antibiotics, driving the metabolic effects.
The new study by NYU Langone Medical Center researchers reveals that mice given lifelong low doses of penicillin starting in the last week of pregnancy or during nursing were more susceptible to obesity and metabolic abnormalities than mice exposed to the antibiotic later in life.
Most intriguing, in a complementary group of experiments, mice given low doses of penicillin only during late pregnancy through nursing gained just as much weight as mice exposed to the antibiotic throughout their lives.
"We found that when you perturb gut microbes early in life among mice and then stop the antibiotics, the microbes normalize but the effects on host metabolism are permanent," says senior author Martin Blaser, MD, the Muriel G. and George W. Singer Professor of Translational Medicine, director of the NYU Human Microbiome Program, and professor of microbiology at NYU School of Medicine. "This supports the idea of a developmental window in which microbes participate. It's a novel concept, and we're providing direct evidence for it."
The researchers stress that more evidence is needed before it can be determined whether antibiotics lead to obesity in humans, and the present study should not deter doctors from prescribing antibiotics to children when they are necessary. "The antibiotic doses used in this study don't mirror what children get," says Laura M. Cox, PhD, a postdoctoral fellow in Dr. Blaser's laboratory and the lead author of the study. "But it has identified an early window in which microbes can influence metabolism, and so further studies are clearly warranted."
In one experiment in the study, Dr. Cox administered water with low doses of penicillin to three groups of mice. One group received antibiotics in the womb during the last week of pregnancy and continued the medication throughout life. The second group received the same dose of penicillin after weaning and, like the first group, continued it throughout life. The third received no antibiotics. "We saw increased fat mass in both penicillin groups, but it was higher in the mice who received penicillin starting in the womb," Dr. Cox says. "This showed that mice are more metabolically vulnerable if they get antibiotics earlier in life."
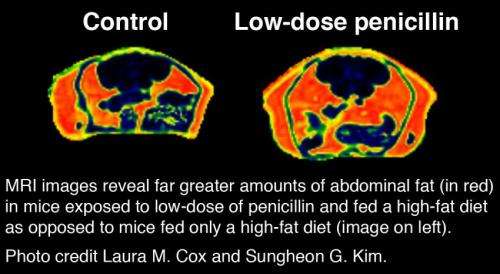
The treated mice also grew fatter than the untreated mice when both were fed a high-fat diet. "When we put mice on a high-calorie diet, they got fat. When we put mice on antibiotics, they got fat," explains Dr. Blaser. "But when we put them on both antibiotics and a high-fat diet, they got very, very fat." Normally, adult female mice carry three grams of fat. The animals in the study fed the high-fat diet had five grams of fat. By comparison, the mice who received antibiotics plus the high-fat chow packed on 10 grams of fat, accounting for a third of their body weight. The treated rodents were not only fatter but also suffered elevated levels of fasting insulin, and alterations in genes related to liver regeneration and detoxification—effects consistent with metabolic disorders in obese patients.
This work confirms and extends landmark research published by Dr. Blaser's lab in 2012 in Nature. That research showed that mice on a normal diet who were exposed to low doses of antibiotics throughout life, similar to what occurs in commercial livestock, packed on 10 to 15 percent more fat than untreated mice and had a markedly altered metabolism in their liver.
Among the unanswered questions in that study was whether the metabolic changes were the result of altered bacteria or antibiotic exposure. This latest study addresses the question by transferring bacterial populations from penicillin-exposed mice to specially bred germ-free, antibiotic-free mice, starting at three weeks of age, which corresponds to infancy just after weaning. The researchers discovered that mice inoculated with bacteria from the antibiotic-treated donors were indeed fatter than the germ-free mice inoculated with bacteria from untreated donors. "This shows us that the altered microbes are driving the obesity effects, not the antibiotics," says Dr. Cox.
Contrary to a longstanding hypothesis within the agricultural world that holds that antibiotics reduce total microbial numbers in the gut, therefore reducing competition for food and allowing the host organism to grow fatter, the team found that the penicillin did not, in fact, diminish bacterial abundance. It did, however, temporarily suppress four distinct organisms early in life during the critical window of microbial colonization: Lactobacillus, Allobaculum, Candidatus Arthromitus, and an unnamed member of the Rikenellaceae family, which may have important metabolic and immunological interactions. "We're excited about this because not only do we want to understand why obesity is occurring, but we also want to develop solutions," says Dr. Cox. "This gives us four potential new candidates that might be promising probiotic organisms. We might be able to give back these organisms after antibiotic treatments."
The researchers worked with six different mouse models over five years to obtain their results. To identify bacteria, they used a powerful molecular method that involves extracting DNA and sequencing a subunit of genetic material called 16S ribosomal DNA. Altogether, the scientists evaluated 1,007 intestinal samples, which yielded more than 6 million sequences of bacterial ribosomal genes, the order of the nucleotides that spell out DNA. Studies like these are possible because of technological advances in high-throughput sequencing, which allows scientists to survey microbes in the gut and other parts of the body. The Genome Technology Center at NYU Langone Medical Center played a key role in identifying the genetic sequences in the study.
More information: Cell, Cox et al.: "Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences., www.cell.com/cell/abstract/S0092-8674(14)00821-6