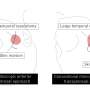
Pigs' hearts transplanted into baboon hosts remain viable more than a year
Investigators from the National Heart, Lung, and Blood Institute (NHLBI) of the National Institutes of Health (NIH) have successfully transplanted hearts from genetically engineered piglets into baboons' abdomens and had the hearts survive for more than one year, twice as long as previously reported. This was achieved by using genetically engineered porcine donors and a more focused immunosuppression regimen in the baboon recipients, according to a study published in The Journal of Thoracic and Cardiovascular Surgery, an official publication of the American Association for Thoracic Surgery.
Cardiac transplantation is the treatment of choice for end stage heart failure. According to the NHLBI, approximately 3,000 people in the US are on the waiting list for a heart transplant, while only 2,000 donor hearts become available each year. For cardiac patients currently waiting for organs, mechanical assist devices are the only options available. These devices, however, are imperfect and experience issues with power supplies, infection, and problems with blood clots and bleeding.
Transplantation using an animal organ, or xenotransplantation, has been proposed as a valid option to save human lives. "Until we learn to grow organs via tissue engineering, which is unlikely in the near future, xenotransplantation seems to be a valid approach to supplement human organ availability. Despite many setbacks over the years, recent genetic and immunologic advancements have helped revitalized progress in the xenotransplantation field," comments lead investigator Muhammad M. Mohiuddin, MD, of the Cardiothoracic Surgery Research Program at the NHLBI.
Dr. Mohiuddin's group and other investigators have developed techniques on two fronts to overcome some of the roadblocks that previously hindered successful xenotransplantation. The first advance was the ability to produce genetically engineered pigs as a source of donor organs by NHLBI's collaborator, Revivicor, Inc. The pigs had the genes that cause adverse immunologic reactions in humans "knocked out" and human genes that make the organ more compatible with human physiology were inserted. The second advance was the use of target-specific immunosuppression, which limits rejection of the transplanted organ rather than the usual generalized immunosuppression, which is more toxic.
Pigs were chosen because their anatomy is compatible with that of humans and they have a rapid breeding cycle, among other reasons. They are also widely available as a source of organs.
In this study, researchers compared the survival of hearts from genetically engineered piglets that were organized into different experimental groups based on the genetic modifications introduced. The gene that synthesizes the enzyme alpha 1-3 galactosidase transferase was "knocked out" in all piglets, thus eliminating one immunologic rejection target. The pig hearts also expressed one or two human transgenes to prevent blood from clotting. The transplanted hearts were attached to the circulatory systems of the host baboons, but placed in the baboons' abdomens. The baboons' own hearts, which were left in place, maintained circulatory function, and allowed the baboons to live despite the risk of organ rejection.
The researchers found that in one group (with a human gene), the average transplant survival was more than 200 days, dramatically surpassing the survival times of the other three groups (average survival 70 days, 21 days, and 80 days, respectively). Two of the five grafts in the long-surviving group stopped contracting on postoperative days 146 and 150, but the other three grafts were still contracting at more than 200 to 500 days at the time of the study's submission for publication.
Prolonged survival was attributed to several modifications. This longest-surviving group was the only one that had the human thrombomodulin gene added to the pigs' genome. Dr. Mohiuddin explains that thrombomodulin expression helps avoid some of the microvascular clotting problems that were previously associated with organ transplantation.
Another difference was the type, strength, and duration of antibody used for costimulation blockade to suppress T and B cell immune response in the hosts. In several groups, longer survival of transplants was observed with the use of anti-CD40 monoclonal antibodies but the longest-surviving group was treated specifically with a high dose of recombinant mouse-rhesus chimeric antibody (clone 2C10R4). In contrast, use of an anti-CD40 monoclonal antibody generated in a mouse (clone 3A8) did not extend survival. Anti-CD40 monoclonal antibodies also allow for faster recovery, says Dr. Mohiuddin.
No complications, including infections, were seen in the longest-survival group. The researchers used surveillance video and telemetric monitoring to identify any symptoms of complications in all groups, such as abdominal bleeding, gastrointestinal bleeding, aspiration pneumonia, seizures, or blood disorders.
The goal of the current study was to evaluate the viability of the transplants. The researchers' next step is to use hearts from the genetically-engineered pigs with the most effective immunosuppression in the current experiments to test whether the pig hearts can sustain full life support when replacing the original baboon hearts.
"Xenotransplantation could help to compensate for the shortage of human organs available for transplant. Our study has demonstrated that by using hearts from genetically engineered pigs in combination with target-specific immunosuppression of recipient baboons, organ survival can be significantly prolonged. Based on the data from long-term surviving grafts, we are hopeful that we will be able to repeat our results in the life-supporting model. This has potential for paving the way for the use of animal organs for transplantation into humans," concludes Dr. Mohiuddin.
More information: "Genetically engineered pigs and target-specific immunomodulation provide significant graft survival and hope for clinical cardiac xenotransplantation," by Muhammad M. Mohiuddin, MD, Avneesh K. Singh, PhD, Philip C. Corcoran, MD, Robert F. Hoyt, DVM, Marvin L. Thomas III, DVM, David Ayares, PhD, and Keith A. Horvath, MD. (DOI: dx.doi.org/10.1016/j.jtcvs.2014.06.002), published in The Journal of Thoracic and Cardiovascular Surgery, Volume 148, Issue 3 (September 2014)