September 10, 2014 report
When spikes collide: Shaking the foundation of neuroscience
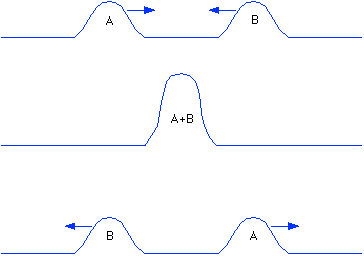
(Medical Xpress)—What happens when pulses on axons collide? Fortunately for neuroscience, that usually only happens when neuroscientists artificially create counter-propagating pulses to study connections. In real brains however, collision is not only an integral reality of every kind neurite, we might imagine it as the principal concern of the neuron itself and the reason they take the unique forms they do. If pulses annihilate upon collision, as the standard theory prescribes, then all might still appear well in Neverland. But if they don't annihilate—that is they pass through each other and continue on—then not only have we misled ourselves about how the structure of a neuron might relate to its activity, but more alarmingly, we have been misled about the fundamental nature of the pulse itself.
In 1949 Ichiji Tasaki studied collision in the myelinated motor fibers innervating the toad sartorius muscle. His conclusions, at least under his particular conditions, were that spikes get annhilated. Tasaki was and still is to neurophysiology what the great Cajal is to neuroanatomy. In other words, he was the man with the plan. If Tasaki found that spikes failed to penetrate each other then that would be good enough for me, and in fact it was good enough for neuroscience for the next half a century.
The reason nobody questioned these findings further is because the Hodkin-Huxley (HH) theory, and the refractory period it so well described, fully accounted for the observed behavior. Refractory periods, we should mention, have been unambiguously described in many experimental nerve preparations. For those familiar with the HH theory, refractory periods (essentially relaxation processes in proteins) are contained in the parametrization of protein conductances. It was attractive at the time to liken the action potential to the burning fuse of gunpowder. In this view even the fastest recovering axon would require some finite time to replenish. If spikes are generated too rapidly, or penetrate from opposite directions, they would encounter a wake of unactivatable channels on both sides.
But Tasaki didn't buy the whole pill, and for that matter neither did the founders of the model. Tasaki rejected the idea that the refractory period is the explanation for spike annhilation. His analysis was that electromechanical events inside and outside the nerve offset each other during collision such that conditions for regenerative pulses are not met. In 1982 he began to explore exactly what this might mean by looking at the mechanical responses of colliding pulses in squid axons.
Taski found that the mechanical pulse was modified at the collision point, but he did not completely examine the details of annhilation itself. Apparently, no other studies have replicated the original 1949 experiments. Falsification of the annhilation scenario really only requires showing sufficient exception, enough that there no longer is a "general case". Thomas Heimburg, from the Neils Bohr Institute in Copenhangen, has recently been able to do just that. Using a simple worm axon preparation where spikes could be timed and measured after stimulation from both ends, Heimburg showed that pulses survive collision.
There is one caveat raised in this. In the ventral cord of the earthworm Lumbricus terrestris, the giant "axon" and the pulse that rides upon it are not the province of any single neuron, but rather collectives that emerge from the six gap-junction linked segmental neurons that knit it together. In other words, each 2ms long pulse is actually a bit longer ithan the full spatial extent of each 1mm length neuron that makes up the axon. For these reasons it was deamed important to demonstrate the collision effects in a more tradional axon. To that end the experiments were repeated in the Lobster Homarus americanus, where the medial giant axons of the ventral cord are each built by a single neuron.
Heimberg recently sent me the new paper that he has on the Arxiv server for review. It is also now forthcoming in Physical Review X. In it he reports that the pulse velocity and shape are unaltered in experimental collisions. Nonlinear hydrodynamics simulations, which showed that soliton-like pulses should penetrate with only minimal amounts of noise generation, were consistent with these observations but inconsistent with refractory period annhilated spikes of the HH model. The energy transmitted in spikes can be associated to different amplitudes and velocities in different kinds of axons. The invertebrate axons used in these studies are considered myelinated but their pulses propagate at speeds much below the 100 m/s upper speed limit for mammalian axons.
There is no reason to presuppose that nature provides for speeds and activation thresholds to be the same in both directions, and also at both ends of any given axon. Furthermore, if nerve diameter varies, we might expect these observable to vary significantly. Of notably intrigue, other researchers have shown that spikes can and do back propagate down an axon from synapse to cell body as part of their normal everyday routine. Indeed these suspicions are among the effects Heimburg noted, with the speed of reverse pulses being slightly slower than the forward. I asked him if he expected that the larger axons might support simultaneous bidirectional penetrating pulses by their possible passage through nonoverlapping domains. His models do not explicity assume or predict any radius dependance for pulse generation or other properties. He noted that membrane domains would be much smaller than pulses and that any inhomogeneities should average. In any event, the experimental evidence for any strict radius dependence of pulse velocity is far from clear.
Where many neuroscientists concern themselves with classifying neurons as say, glutamatergic or dopaminergic, Heimburg might have them all be 'solitonergic'. By that one would mean they all propagate pulses as solitary acoustic waves at a lipid interface. Establishing any new dogma typically requires, among other many intangibiles, demonstration in multiple convincing ways. A nice next step might be looking these phenomena in the canonical Mauthner cell-mediated C-start reflex of the fish. The well-characterized Mauther cell spreads it influence far and near throughout the fish brain and the thoughtful spikes it produces take into account diverse electromagnetic and ephaptic influences. Furthermore the neurites of these diffuse cells undoubtedly hold local conversations in near isolation of each other, yet appear capable of hushing on demand to carry more important organismal concerns when needed.
In such a setup as this, the difference between sending spikes in different regions and directions that either collide or don't is profound. Understanding these potential limits or surfeits of information processing in neurites is critical.
More information: Penetration of action potentials during collision in the median and lateral giant axons of invertebrates, arXiv:1404.3643 [physics.bio-ph] arxiv.org/abs/1404.3643
© 2014 Medical Xpress


















