April 18, 2014 report
Know the brain, and its axons, by the clothes they wear
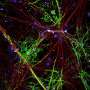
(Medical Xpress)—It is widely know that the grey matter of the brain is grey because it is dense with cell bodies and capillaries. The white matter is almost entirely composed of lipid-based myelin, but there is also a little room in the grey matter for a few select axons to be at least partially myelinated. A group of well known researchers, mostly from Harvard and MIT, decided to look for possible patterns in the myelin found in cortical grey matter. Their Science published findings suggest that this dynamic balance struck up by each axon, somewhere between zero and full myelination, does not tip to the benefit of action potential speed alone. Instead, it follows a more subtle give and take between different kinds cells.
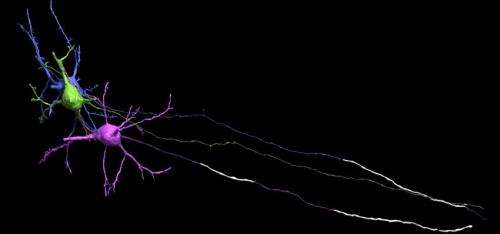
In looking down the length of an axon, longitudinally that is, each segment of myelin is separated by a node. The thickness of the myelin coat varies significantly from node to node. Presumably then, so does the speed and reliability of the spike propagated in that segment. The researchers suggest however, that it is more the phase and offset of these nodes that matters. The distance to first node in particular is important because it is here that the spike shape is first initiaillized. As Doug Fields points out in a perspective that accompanies the paper, spike shape (usually inconsequential in computational models) has important functional implications including the amount of transmitter released, the refractory period and the spike frequency.
Within the cortical grey, it is now known that the bare initial segment of the axon is irresistible to other cells. Their synaptic overtures are regularly accepted and also reciprocated by the axon's own transmitter release from bare, noncanonical release sites. The researchers found that the length of the myelin-free axon initial segment had a graded distribution with the more superficially located pyramidal cells in the mouse cortex having longer "open" axon. In layer II/III bare stretches up to o 55 µm were evident.
The technology that makes it possible to reconstruct serial sections of brain is perhaps the most advanced—and certainly the most industrialized—in neuroscience. It is precisely the same technique used in the recent Brainbow II studies, which incidentally have also yielded some the most celebrated images in science. But I must say, reader, if you are not blown away by the above mentioned details on myelination, you are not alone. That you are still here indicates that you expect something more.
So forgive me, rightfully esteemed authors, if I suggest you have an opportunity here perhaps not yet missed, but rapidly growing stale. Ken, Sebastian, Jeff—Doug, where is the missing myelin mechanics? In the name of all that is Holy, myelination requires a breaking of symmetry, namely it has to wrap in one direction. We have asked previously, in detail, how this constraint is applied in whole brain and nerve, going down an axon, going to immediately adjacent axons, and also to the multiple arms of any one oligodendrocyte.
As myelin undergoes phase transitions in development, does its 3D tubular mesh align like slow motion lipid spin glasses? Is direction imposed individually at each turn, or in bulk transition, perhaps reflective of temperature dependent crystal or magnetic domain formation? More speculatively, can firing axons, simultaneously pulsing mechanically in the radial direction, rectify their continuous cellular substructure into miniscule torques which aid and abet myelination? How does bulk myelination vary across the bilaterally symetric halves of the brain, across the callosum, and down the altogether unique myelin of the nerves units of the body? Now that we clearly have the technology, lets answer these questions and begin to piece this brain together ground up.
The power of the screw and the drill, known to any machinist, is not lost here. The authors own recent incredible work attests to that. They reference their previous discovery of helical substructure in stacked endoplasmic reticulum sheets connected through unique membrane motiffs. Might neurons themselves be chiral, or at least their axons or apical dendrite have a preferred hand? If it is now possible to image effervescent cell organelles, centriolar-defined coordinate systems, the windings of microtubule arrays even down to the tiny symmetry-breaking protein hooks which preferentially adorn them in axons vs dendrtites, certainly we can now construct geometry on larger scales of the brain.
More information: Paper: Myelin—More than Insulation, Science 18 April 2014: Vol. 344 no. 6181 pp. 264-266. DOI: 10.1126/science.1253851
Perspective: http://www.sciencemag.org/content/344/6181/264.full
© 2014 Phys.org