Team discovers lung regeneration mechanism
.jpg)
A research team led by Jackson Laboratory Professors Frank McKeon, Ph.D., and Wa Xian, Ph.D., reports on the role of certain lung stem cells in regenerating lungs damaged by disease.
The work, published Nov. 12 in the journal Nature, sheds light on the inner workings of the still-emerging concept of lung regeneration and points to potential therapeutic strategies that harness these lung stem cells.
"The idea that the lung can regenerate has been slow to take hold in the biomedical research community," McKeon says, "in part because of the steady decline that is seen in patients with severe lung diseases like chronic obstructive pulmonary disease (known as COPD) and pulmonary fibrosis."
Nevertheless, he notes, there are examples in humans that point to the existence of a robust system for lung regeneration. "Some survivors of acute respiratory distress syndrome, or ARDS, for example, are able to recover near-normal lung function following significant destruction of lung tissue."
Mice appear to share this capacity. Mice infected with the H1N1 influenza virus show progressive inflammation in the lung followed by outright loss of important lung cell types. Yet over several weeks, the lungs recover, revealing no signs of the previous lung injury.
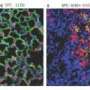
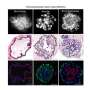
Using this mouse model system, McKeon and his colleagues had previously identified a type of adult lung stem cell known as p63+/Krt5+ in the distal airways. When grown in culture, these lung stem cells formed alveolar-like structures, similar to the alveoli found within the lung. (Alveoli are the tiny, specialized air sacs that form at the ends of the smallest airways, where gas exchange occurs in the lung.) Following infection with H1N1, these same cells migrated to sites of inflammation in the lung and assembled into pod-like structures that resemble alveoli, both visually and molecularly.
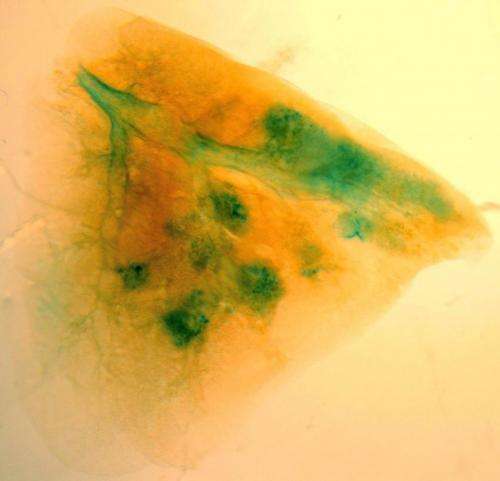
In the new paper, the research team reports that the p63+/Krt5+ lung stem cells proliferate upon damage to the lung caused by H1N1 infection. Following such damage, the cells go on to contribute to developing alveoli near sites of lung inflammation.
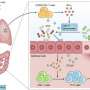
To test whether these cells are required for lung regeneration, the researchers developed a novel system that leverages genetic tools to selectively remove these cells from the mouse lung. Mice lacking the p63+/Krt5+ lung stem cells cannot recover normally from H1N1 infection, and exhibit scarring of the lung and impaired oxygen exchange—demonstrating their key role in regenerating lung tissue.
The research team also showed that when individual lung stem cells are isolated and subsequently transplanted into a damaged lung, they readily contribute to the formation of new alveoli, underscoring their capacity for regeneration.
In the U.S. about 200,000 people have ARDS, a disease with a death rate of 40 percent, and there are 12 million patients with COPD. "These patients have few therapeutic options today," Xian says. "We hope that our research could lead to new ways to help them."
More information: Zuo et al.: "p63+/Krt5+ distal airway stem cells are essential for lung regeneration." Nature, 11/12/14, DOI: 10.1038/nature13903