February 23, 2015 report
Stopping plaque formation in arteries using amphiphilic nanoparticles
Heart disease is one of the leading causes of death among Americans. Hardening of the arteries, or atherosclerosis, is one form of heart disease, but can also occur in other arteries within the body. Standard therapy for this is either insertion of a stent or taking cholesterol-lowering drugs, which manages the disease, but does not block the actual mechanisms that cause plaque buildup.
Lead authors Daniel R. Lewis and Latrisha K. Peterson from Rutgers University developed an approach that, in mouse studies, effectively stopped the early buildup of fatty plaque in the artery and halted disease progression using amphiphilic nanoparticles. Their work appears in the Proceedings of the National Academy of Sciences.
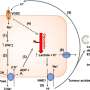
There are several steps that lead to atherosclerosis. An artery has an endothelial cell monolayer that is in contact with the blood. When this layer is inflamed due to the presence of oxidized low-density lipoprotein (LDL), white blood cells, or macrophages, are recruited as cellular housekeepers to engulf oxidized LDL. They do this by expressing scavenger receptors. Once the lipid is engulfed by the macrophage, a foam cell, named for its "foamy" appearance, is formed. Macrophages not only form foam cells, but it also activates cytokines which cause inflammation, perpetuating the process by signaling for more macrophages.
Arteries harden from smooth muscle cells moving into the intima, combing with collagen, forming a lesion. It these lesion sites where more plaque forms and atherosclerosis can progress.
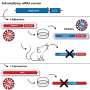
Lewis and Peterson et al. developed a general method for selecting a sugar-based amphiphilic macromolecule with properties that allows to selectively bind to oxidized LDL. These macromolecules were then complexed around a hydrophobic core to form serum-stable nanoparticles so that they can traverse through the body to sites of greatest plaque buildup. The intention is stop the signals that turn on consumption of oxidized LDL and subsequent formation of foam cell as well as stop inflammation.
When screening for the best candidates for an amphiphilic molecule, they looked for three properties:
- Their molecule needs to bind to the scavenger receptors, so that they will accumulate near the lesion, or places of greatest plaque buildup.
- Their molecule needs to out-compete oxidized LDL uptake, thus preventing foam cell formation
- Their molecule needs to down regulate the cell surface expression of the scavenger receptors so more white blood cells are not recruited to the site.
To accomplish this, they designed macromolecules with similar chemical features as oxidized LDL to counteract oxidized LDL and scavenger receptor binding. Stereochemistry played a large role in the binding process.
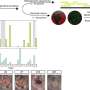
After testing the various candidates, M12PEG was most effective in both binding to the scavenger receptor and halted the signals for recruiting more white blood cells. They then made amphiphilic core/shell nanoparticles using flash nanoprecipitation. Mouse studies showed that the nanoparticle remained within the mice long enough to distribute throughout the body. Additional studies showed that nanoparticles tended to collect in places with the greatest plaque buildup, which were the aortic arch and carotid branch points. Finally, in mice treated with the nanoparticles, their lesions were less developed and they had 37% less artery blockage compared to untreated mice, indicating that the nanoparticles stopped disease progression.
Lewis and Peterson state the results indicate that "following penetration into the atherosclerotic plaques, the [nanoparticles] bind scavenger receptors of plaque-resident cells and prevent oxidized lipid-cellular interactions. By decreasing lipid accumulation within the artery, local inflammation would be lower and result in an overall reduction in artery occlusion."
Overall, the amphiphilic nanoparticles worked better than prior studies with micelles to selectively bind to the scavenger receptors preventing the consumption of oxidized LDL and subsequent foam cell formation. Additionally, they halt the progress of atherosclerosis by down regulating scavenger receptor expression. Furthermore, Lewis and Peterson et al. present a method for screening molecules that can be used to as a computational model for drug development.
More information: "Sugar-based amphiphilic nanoparticles arrest atherosclerosis in vivo". DOI: 10.1073/pnas.1424594112
© 2015 Medical Xpress