March 24, 2015 report
How to myelinate a nerve
(Medical Xpress)—Demyelinating diseases, like MS or ALS, attack an otherwise healthy nervous system and leave its owner a prisoner in their own body. The harder big pharma searches for—and fails to find—miracle drugs to cure these diseases, the more obvious a single fundamental truth becomes: drugs may alleviate some symptoms for a little while, but to fix these diseases we need to know what it takes physically to remyelinate axons. To do that, we need to know how they are myelinated in the first place. Many neuroscientists are now studying various molecular pathways that appear to be critical for myelination. Unfortunately they don't always tell us much about how axons are myelinated. For that, we need to watch it happen in slow-mo.
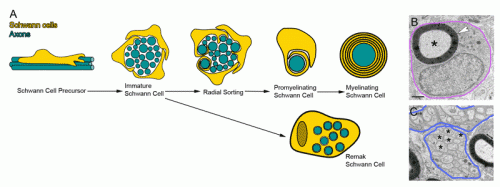
A recent review in Developmental Cell titled 'Dynamics and Mechanisms of CNS Myelination' takes stock of the current state of the art in visualizing what is actually going on here. Although theories abound with colorful names like the 'liquid mosaic,' 'jelly roll,' and 'carpet crawler' models, a clear consensus as to which is correct has yet to emerge. A complementary study in Neuron elaborates on what may be the central issue for Schwann cell myelination in the PNS. Namely, how do these cells tackle the problem of radial sorting. In other words, after a few promyelinating Schwann cells corral a group of potential axon partners into their mutual interior, how do they then switch gears, match up, and establish a mature pattern of of one-to-one myelination?
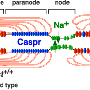
The authors focused on the role of the extracellular matrix in kicking off the radial sorting process. Schwann cells themselves are in part responsible for secreting the various laminins, collagens, and heparin sulfate proteoglycans that make up the matrix. One of the main triggers to start secretion is a critical level of spike activity in the axons. The authors looked the role of a newly defined family of receptors known as aGPCRs (adhesion G protein-coupled receptors). As a more general class, the GPCRs are among the oldest receptor proteins found in nervous systems.
In particular the authors were able to establish the importance of the large N-terminal extracellular domains of these proteins in interacting with the basal lamina during radial sorting. This was done mainly by creating various mutants and looking at high resolution EM images. Getting the makeup of the matrix right is just one small piece of the puzzle. To actually watch myelination as it happens you need time lapse fluorescence microscopy and the right labels on the things you want to see. Sue Barnett, from the University of Glasgow has been able to do this using ex vivo preparations of mouse spinal cord.
Her group reported that once oligodendrocytes in the CNS get their axon, they spiral around it in a corkscrew fashion. After that there is focal expansion of these processes into short longitudinally extending cuffs which then overlap each other. They call their model, the ''ofiomosaic model'' (o´Qiz+mvsa¨iko´ is Greek for snake + mosaic), but words can't really do it justice. They say it is a combination of a previous patchwork quilt model and a serpent model, but as you can probably tell, you have to see it with your own eyes to believe it.
A spiraling corkscrew process is intriguing because it necessarily has a handedness or polarity. Potentially that is a critical constraint which could influence how adjacent myelin segments interact and organize themselves. It is also interesting in light of recent studies showing definite handedness to spiral preferences in extending axons and their filopodia. I asked Sue if they saw any indication of directional preferences in their studies, and if so what was the preferred handedness of spiral myelination. She said that at this point it is difficult to tell, and also difficult to image the whole oligodendrocyte. One interesting thing they did notice, which classically isn't supposed to happen at all, was the presence of 'two myelin internodes' on an axon in vitro.
The detailed structure of myelinated nodes, paranodes, and internodes is now coming into full view. Understanding what makes these unique membrane elements work, and the idosyncratic forms they take both in the PNS and the CNS, are the first steps towards getting them to reform in patients with demyelinating disease.
More information: — Dynamics and Mechanisms of CNS Myelination, Developmental Cell, www.cell.com/developmental-cel … /abstract/S1534-5807%2815%2900059-3
The Adhesion GPCR GPR126 Has Distinct, Domain-Dependent Functions in Schwann Cell Development Mediated by Interaction with Laminin-211, Neuron, www.cell.com/neuron/abstract/S0896-6273(14)01166-0
© 2015 Medical Xpress