November 21, 2014 report
Built for speed: paranodal junction assembly in high performance nerves
(Medical Xpress)—The nervous system is host to some of the most sophisticated cellular structures found anywhere in the body. The orderly phalanxes of sterocilia which arm the ear's inner hair cells, and the photosensitive dishes stacked into precision-built lipid cup boards in cones of the eye are two fine examples of structures whose function is well understood. An equally intriguing, and perhaps even more elaborate cellular creation, is the uniquely geometric front that is established on the borderland between two myelinating cells that wrap adjoining segments of an axon. In a recent paper in Nature Neuroscience, researchers describe the critical role played by molecules known as ankyrins in the assembly of axoglial junctions, and reveal the surprising origins of these cytoskeletal scaffold proteins.
Like many proteins, the expression of ankyrins (the name comes from the Greek word for fused), is fiendishly complex. They are transcribed from several different promoters, and undergo extensive alternative splicing after transcription. Fortunatly much of these events are well understood. Less clear, at least as far as the axoglial junction is concerned, is which cells make them, which cells actually employ them, and where. It was previously known that neurons make two forms of ankyrin, AnkG and AnkB, which together regulate the formation of the axon initial segment. Notably, these proteins distribute and cluster the ion channels in that region to control the generation of spikes.
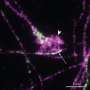
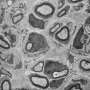
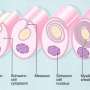
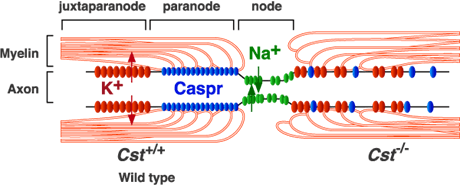
The so-called nodes of Ranvier found between any two adjacent myelin segments are generally divided into three regions: the node's eye at the center; the paranode around it where glial loops form their signature septate junctions; and the juxtaparanodal area just outside the paranodes. In myelinated axons at least, AnkG and AnkB has been found beyond the initial segment, primarily in the vicinity of the nodes. It was therefore presumed that these proteins were delivered by the axon transport system to these locations.
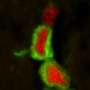
The new research just presented in Nature says this presumption was wrong. A closer look revealed that these proteins are actually expressed by the myelinating cells and are highly enriched on the glial side of the paranodal junction. AnkB was found in the Schwann cells of the peripheral nervous system while AnkG was the go to ankyrin for the oligodendrocytes of the CNS. Supporting this conception was the finding that when AnkG was conditionally knocked out in mouse oligodendrocytes, the formation of paranodal junctions were disrupted, and nerve conduction was correspondingly delayed during development.
Knockouts of any one protein are often revealing, but not always definitive. For example, when the researchers knocked out AnkB in dorsal root ganglion neurons, but not Schwann cells, the paranodes still looked fine in their culture. While this similarly suggests a Schwann cell source of AnkB in the live animal state, there is always the possibility that there are always multiple cellular sources of the protein, or even that its absence in axons is sensed by other cells which in turn upregulate it.
Ankrins are closely related to the spectrins, which are critical in red blood cell structure. It has recently been shown that some spectrins form alternating bands with actin in axons. While these bands, or rather rings, do not show the crystalline order of actins muscle tissue, this discovery suggests that axons have an internal order that extends significantly beyond the node region. While ankyrins provide one good lens to view node structure, there are plenty of other equipotent proteins that need to be considered in trying to generate a conceptual microcosm of the node.
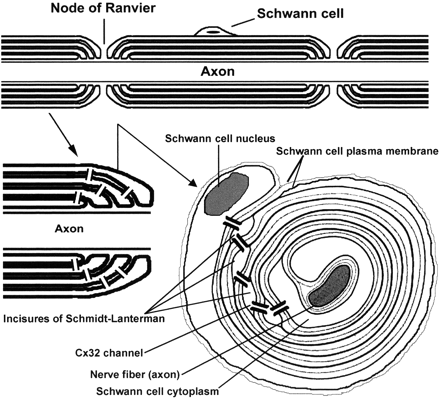
For example, there are several gap junction proteins expressed in the node region which are critical in providing bypass channels directly through the multple layes of myelin. One such protein, connexon 32, has been localized exclusively to the diagonally-arranged Schmidt–Lanterman incisures. These small interior cytoplasmic swellings are similar to the paranodal loops, only they are found in the center portion of the myelin segment. Dye-transfer experiments on connexon 32 knockout mice indicate that other gap junctions must be present. In other words, when dye was pumped into the body of the myelinating cell, it was still conducted directly through the patent incisures to the axon as easily as a soul would traverse the tunnels through the interior of the Great Pyramid.
While researchers have theoretically linked these tunnels to potassium or other cation flows, perhaps it is more instructive to look at a broader range of their possible physics. The link from structure to function is often best perceived through the dynamic aspects of development. Here we would be seeking the inferred physical forces, the hydraulic flows that would be conducted in either direction between the leading edge compartments of the myelin membrane as it actively wraps the axon, and the exterior reservoir. The rate of growth of myelin is known to be dependant on the spiking activiting of the encircled axon. The most direct and efficient coupling of energy from spike to myelin may be found in the tiny displacements and temperature/phase transitions that are transduced or otherwise rectified into torques which influence myelination direction and magnitude.
Rather than looking at the node as an elaborate insulating apparatus that enhances electrical aspects of the spike, we might view its structure more as the result and signature of the mechanical output of the axon, and ultimately a contributer and refiner of it. In this view, the now established acoustic or vibrational aspects of the spike might begin to be absorbed into a more global view of axoglial performance. As more detailed imaging studies with a big data bent turn their scrutizing eye to glial cells, the constraints we need for this kind of theory will be in hand. Many such constraints are simple observables, perhaps aided with special labels: What direction is the myelin in adjacent and successive segments down the axon, in adjacent axons of a nerve, and for oligodendrocytes, for the multiple arms of myelin they elaborate?
Similarly, what is the physical origin of the one myelinating arm of the Schwann verses the many of the oligodendrocyte - is it just physical size and resource allocation? If so can that explain why Shwann cells shoot of numerous microvilli into the node region whereas the CNS instead relies on the supplementary feet of special astrocytics (called perinodal processes) to do this job for oligodendrocytes?
One hint may come from the observation that much of our understanding of development comes from asking the question, what else in the body is some particular feature like? For Schwann cells, one suggestion is that they are like modified epithelial cells in the organization of their connective and junctional proteins, and also in their orientation relative to the extracellular basal lamina. If that is in any sense a true metaphor than already we know much about them at the molecular control level.
More information: Glial ankyrins facilitate paranodal axoglial junction assembly, Kae-Jiun Chang Daniel R Zollinger Keiichiro Susuki Diane L Sherman Michael A Makara Peter J Brophy Edward C Cooper Vann Bennett Peter J Mohler & Matthew N Rasband, Nature Neuroscience aop, (2014) DOI: 10.1038/nn.3858
© 2014 Medical Xpress