Effects of n-3 PUFAs on insulin sensitivity unclear
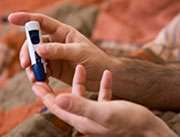
(HealthDay)—Dietary n-3 polyunsaturated fatty acids (PUFAs), including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), do not appear to have clinically meaningful effects on peripheral or hepatic insulin sensitivity in insulin-resistant adults without diabetes, according to research published online April 7 in Diabetes Care.
Antigoni Z. Lalia, M.D., of the Mayo Clinic College of Medicine in Rochester, Minn., and colleagues conducted a randomized, double-blind study in which 31 insulin-resistant adults, without diabetes, received either 3.9 g/day EPA+DHA or placebo for six months. A hyperinsulinemic-euglycemic clamp with somatostatin was used to assess hepatic and peripheral insulin sensitivity. Insulin secretion and muscle mitochondrial function were also evaluated.
The researchers found that, compared with placebo, EPA+DHA did not affect peripheral insulin sensitivity, postprandial glucose disposal, or insulin secretion. A small, but significant, improvement in hepatic insulin sensitivity, determined from the suppression of endogenous glucose production by insulin, was observed for EPA+DHA compared with placebo. Neither EPA+DHA nor placebo influenced muscle mitochondrial function.
"This study demonstrates that dietary EPA+DHA does not improve peripheral glucose disposal, insulin secretion, or skeletal muscle mitochondrial function in insulin-resistant nondiabetic humans," the authors write. "There was a modest improvement in hepatic insulin sensitivity with EPA+DHA, but this was not associated with any improvements in clinically meaningful outcomes."
One author is a member of the Scientific Advisory Board of Sancilio and Company, which supplied materials for the study.
More information:
Abstract
Full Text (subscription or payment may be required)
Copyright © 2015 HealthDay. All rights reserved.