Molecular mechanism for regulating blood sugar could enhance understanding of diabetes

A key mechanism in the regulation of blood sugar levels has been identified in work led by A*STAR researchers. The findings could help us understand the mechanisms of diabetes and might lead to new treatments.
Blood levels of the sugar glucose increase after a meal. In response, the body releases insulin, which activates mechanisms to return these levels to normal. One such mechanism involves transport of the protein GLUT4 to the membranes of muscle and fat cells, enabling the uptake of glucose from the blood into the cells.
"Defects in glucose uptake in response to insulin generally manifest as type 2 diabetes," explains Han Weiping from the Laboratory of Metabolic Medicine at A*STAR Singapore Bioimaging Consortium who led the study. "Insulin-stimulated GLUT4 translocation is central to the maintenance of blood glucose levels. An understanding of the mechanisms that underlie this process will help us to work out what goes wrong in diabetes."
Previous studies had identified two important elements involved in the transport of GLUT4: an insulin-activated kinase called Akt2 and rearrangement of the cell's skeleton, which consists of fibers of the protein actin. Han, his graduate student Lim Chun-Yan and collaborators investigated the molecular pathway that connects these two elements.
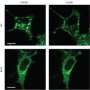
They did this by screening proteins in fat cells to identify those involved in the delivery of GLUT4 to cell membranes. They identified one—tropomodulin3 (Tmod3)—that is also involved in the rearrangement of the cell's actin skeleton. The researchers showed that Tmod3 is modified by insulin-activated Akt2 and that GLUT4 translocation and glucose uptake are impaired in cells with reduced Tmod3 levels.
By making cells with fluorescently labeled actin, the team also found that low expression of Tmod3 reduced the ability of a cell to rearrange its skeleton in response to insulin; the same effect was seen in cells that expressed a form of Tmod3 that cannot be modified by Akt2. The fluorescently labeled actin also revealed that modification of Tmod3 by Akt2 potentiates the insulin-induced actin reorganization.
"Our study highlights a direct link between Akt2 signaling and the actin skeleton that is essential for insulin-stimulated GLUT4 translocation," says Han. "But the identification of Tmod3 might represent the tip of the iceberg of a vast signaling network involved in glucose uptake. A complete understanding of the mechanism of GLUT4 transport would help to pinpoint the key molecular nodes that underlie this process and to develop a therapeutic intervention to help clear glucose more efficiently from the body."
More information: "Tropomodulin3 is a novel Akt2 effector regulating insulin-stimulated GLUT4 exocytosis through cortical actin remodeling." Nature Communications 6, 5951 (2015). dx.doi.org/10.1038/ncomms6951