Team develops novel gene therapy for achromatopsia
Researchers from the University of Pennsylvania and Michigan State University presented new preclinical data this week that evaluates the efficacy of a gene therapy treatment for achromatopsia, a rare inherited retinal disease that involves cone cells. The disease affects humans as well as dogs.
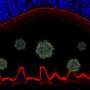
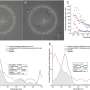
Trying the treatment in dogs with a mutation in a gene that causes achromatopsia, the treatment demonstrated a functional rescue of cone cells in nearly 100 percent of treated eyes.
Study results were presented in an oral session at the Association for Research in Vision and Ophthalmology Annual Meeting in Denver.
The research was co-led by Penn's Gustavo Aguirre, a professor of medical genetics and ophthalmology at the School of Veterinary Medicine, and András Komáromy, an associate professor at Michigan State's College of Veterinary Medicine.
"We are excited to contribute additional evidence suggesting that investigational gene therapies for treating inherited eye disorders show a high degree of potential," Aguirre said.
The investigational gene therapy was developed by Applied Genetic Technologies Corporation, a clinical stage biotechnology company.
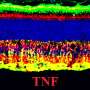
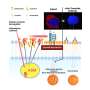
The study evaluated the efficacy of an experimental viral vector, known as AAV, engineered to express the human cyclic nucleotide gated channel beta 3 (hCNGB3) gene, in restoring function to cone cells in the retinas of dogs affected by achromatopsia. Cone cells are specialized cells primarily responsible for color vision.
The study assessed the efficacy of AAV vectors expressing hCNGB3 or codon-optimized hCNGB3 cDNAs, driven by two versions of the human red cone opsin promoter and packed in one of three types of AAVs. Results demonstrated that functional rescue of cone cells was observed in nearly 100 percent of eyes treated with five combinations of the viral vectors, opsin promoters and restored gene.
"This is an important advancement, especially given the lack of available therapeutic options," said Komáromy.
Achromatopsia is an inherited retinal disease characterized by complete loss of cone photoreceptor function, and people with achromatopsia have severe vision loss that renders them legally blind. The disease can be caused by mutations in several genes, with mutations in CNGB3 accounting for half of all cases in Western countries; there is currently no cure.
This gene therapy product, currently in development, aims to restore cone function.
More information: www.arvo.org/webs/am2015/abstract/268.pdf