Genetic variation determines response to anti-diabetic drug
In the first study of its kind, Penn researchers have shown how an anti-diabetic drug can have variable effects depending on small natural differences in DNA sequence between individuals. Mitchell Lazar, MD, PhD, Raymond Soccio, MD, PhD, and colleagues at the Perelman School of Medicine at the University of Pennsylvania, aim to apply this knowledge to develop personalized approaches to treating diabetes and other metabolic disorders. The team published their findings this week in Cell.
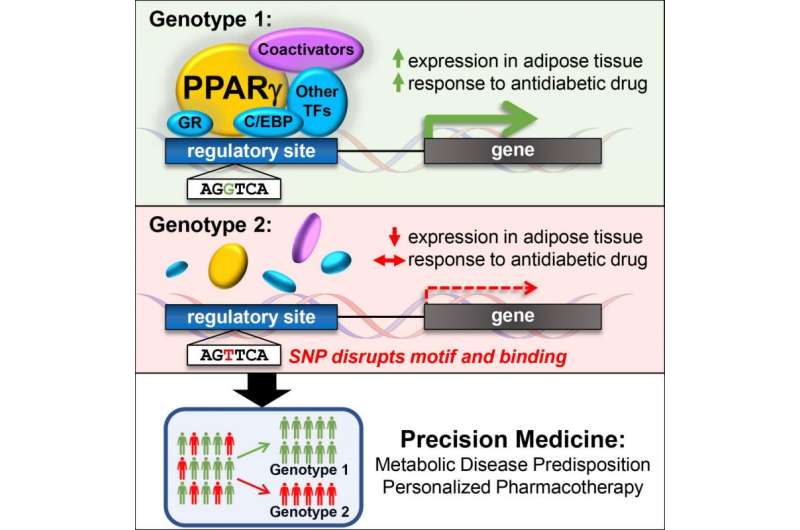
Lazar and team focused on the important fat cell molecule called PPAR-gamma, which is the target of the thiazolidinedione (TZD) class of drugs used to treat type 2 diabetes. PPAR-gamma binds to DNA at switches that turn other genes on, and the Penn researchers showed that natural genetic differences in the DNA of this regulatory switch could determine whether PPAR-gamma and TZD drugs could turn other genes on. "The implications of this work go beyond PPAR-gamma and TZDs, to all drug targets that function directly at the genome to regulate physiology in health and disease," said Soccio, the study's lead author and an instructor in Medicine. Indeed, 20 percent of all prescriptions written are for such drugs as thyroid hormone and steroids that target nuclear receptor proteins related to PPAR-gamma, noted senior author Lazar, professor of Medicine & Genetics and director of the Institute for Diabetes, Obesity, and Metabolism.
The genetic differences are called SNPs, or single nucleotide polymorphisms, and are variants in the DNA alphabet of A, T, C, and G molecules that occur naturally among individuals. Many such SNPs have been associated with disease risk, for instance showing that a person with an A at a given location in DNA has a higher risk of diabetes compared with someone with a G. However, these disease-related SNPs often reside in the so-called "dark matter" of the genome that does not directly code for genes, but includes those switches that control genes.
Lazar and his team showed that SNPs in PPAR-gamma switches provide a mechanism for the disease-risk associations. For example, one such SNP was linked to blood lipids, including HDL ("good" cholesterol) and triglycerides; type 2 diabetes; high blood pressure; and waist-hip ratio (a measure of "apple" versus "pear" body shape in obesity). This constellation of findings is called "the metabolic syndrome", and Soccio points out that "it's remarkable that a single change in a DNA letter determines whether PPAR-gamma binds to one regulatory site in fat tissue, and this may alter a person's risk of metabolic syndrome."
The Penn team started with animal studies, showing that natural SNPs differing between mouse strains could determine whether PPAR-gamma finds its way to its DNA regulatory switch in the dark matter of the genome. Next, they treated mice with TZDs and found that these SNPs could also determine whether the drug turned genes on.
The fact that an individual's genetic makeup can affect drug response has led to the relatively new field of personalized pharmacogenomics. "Our study provides proof-of-concept that naturally occurring regulatory genetic variation can affect nuclear receptor-mediated gene activation and, more generally, drug response in living animals," Lazar said. "This has special significance for TZDs, which have powerful anti-diabetic effects but limited clinical utility due to non-response, side effects, and adverse events."
TZDs are the only diabetes drugs that target fat cells and enhance a diabetic's own response to insulin, yet they have recently fallen into disuse due to side effects like edema and bone loss, as well as reported risks of heart attacks and bladder cancer. What's more, 20 percent of patients with type 2 diabetes fail to improve their diabetes control on TZDs.
"One day the approaches we used in this study can be used predict who will benefit most from TZD-like drugs, so now we need to determine the pattern of SNP differences that together may show why these drugs have benefits or harms for one person and not another," explains Soccio. "This type of precision medicine is a major goal of our research efforts."
The team moved on to study human fat tissue, provided by obese bariatric surgery patients at Penn Medicine. Similar to mice, variation in human SNPs also affected PPAR-gamma binding to DNA regulatory switches, allowing the researchers to "interrogate" these SNPs in powerful large human genetic studies. They used a Finnish database to find how many genes are "on" in fat tissue from over 1,500 people to show that SNPs affecting PPAR-gamma binding also determined gene activity levels in the human population. Finally, they interrogated large human genetic analyses called genome-wide association studies (GWAS) and found that SNPs affecting PPAR-gamma binding in fat underlie some of the risk for metabolic diseases.
These studies show for the first time that natural genetic variation in PPAR-gamma binding to DNA in fat cells can determine individual disease risk and drug response. "Our work shines a bright light on genetic differences in PPAR-gamma bound regulatory DNA, and how these differences among people may allow us to better predict metabolic disease and then apply precision medicine for treatment or prevention," Soccio said.