Study finds mechanism that turns white fat into energy-burning brown fat
Columbia University Medical Center (CUMC) researchers have identified a mechanism that can give energy-storing white fat some of the beneficial characteristics of energy-burning brown fat. The findings, based on studies of mice and of human fat tissue, could lead to new strategies for treating obesity and type 2 diabetes. The study was published today in the online edition of the journal Cell.
Humans have two types of fat tissue: white fat, which stores excess energy in the form of triglycerides, and brown fat, which is highly efficient at dissipating stored energy as heat. Newborns have a relative abundance of brown fat, as protection against exposure to cold temperatures. In adults, however, almost all excess energy is stored as white fat.
"Turning white fat into brown fat is an appealing therapeutic approach to staunching the obesity epidemic, but it has been difficult to do so in a safe and effective way," said study leader Domenico Accili, MD, professor of Medicine and the Russell Berrie Foundation Professor at CUMC.
White fat can be "browned" with a class of drugs called thiazolidazines (TZDs), which increase the body's sensitivity to insulin. However, TZDs have many adverse effects — including liver toxicity, bone loss, and, ironically, weight gain — which have limited the use of these drugs.
The current study was undertaken to learn more about the function of TZDs, with the ultimate goal of developing better ways to promote the browning of white fat.
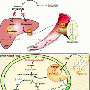
Scientists have known that TZDs promote the browning of white fat by activating a cell receptor called peroxisome proliferator-activated receptor–gamma (ppar-gamma), but the exact mechanism was not clear. To learn more, Dr. Accili and his colleagues studied a group of enzymes called sirtuins, which are thought to affect various biological processes, including metabolism.
The researchers had previously shown in mice that when sirtuin activity increases, so does metabolic activity. In the present study, they found that sirtuins boost metabolism by promoting the browning of white fat. "When we sought to identify how sirtuins promote browning, we observed many similarities between the effect of sirtuins and that of TZDs," said lead author Li Qiang, PhD, associate research scientist in Medicine at CUMC.
Sirtuins work by severing the chemical bonds between acetyl groups and proteins, a process known as deacetylation. "So the next question was whether sirtuins remove acetyl groups from ppar-gamma and, indeed, that was what we found," said Dr. Qiang.
To confirm that the deacetylation of ppar-gamma is crucial to the browning of fat, the researchers created a mutant version of ppar-gamma, in effect mimicking the actions of sirtuins. The mutation promoted the development of brown fat–like qualities in white fat.
"Our findings have two important implications," said Dr. Accili. "First, they suggest that TZDs may not be so bad — if you can find a way to tweak their activity. Second, one way to tweak their activity is by using sirtuin agonists — that is, drugs that promote sirtuin activity."
"The truth is, making sirtuin agonists has proved to be a real bear — more promise than fact," he continued. "But now, for the first time, we have a biomarker for good sirtuin activity: the deacetylation of ppar-gamma. In other words, any substance that deacetylates ppar-gamma should in turn promote the browning of white fat and have a beneficial metabolic effect."
More information: "Brown Remodeling of White Adipose Tissue by SirT1-Dependent Deacetylation of Ppar-gamma." Cell, 2012.