A protein vaccine designed to target all four variations of the dengue virus proves effective in mouse trials
A breakthrough in the search for safe immunization against dengue fever has emerged after trials at A*STAR showed a new vaccine without live viruses induces an effective immune response.
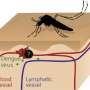
Dengue fever is a debilitating tropical disease spread by mosquitoes, against which a global research effort has failed to develop a universally approved vaccine. Crucially, the dengue virus comes in four variations, known as serotypes, and for a vaccine to be effective it must target all four serotypes at once. Cuban scientists developed a new potential vaccine and sought input from Katja Fink and colleagues at the A*STAR Singapore Immunology Network (SIgN).
"Several dengue vaccine candidates have been developed, but they all have shortcomings," says Fink. "Vaccines that contain live viruses can produce a strong immune response, but cause unwanted side effects. From a safety point of view it is preferable to have protein-only approaches."
One promising candidate is the dengue virus E protein, which includes epitopes—protein regions that are recognized and targeted by the immune system—found on all four dengue serotypes. A specific part of the E protein, called DIII, is targeted by particularly potent neutralizing antibodies. However, DIII tends to attract only one type of lymphocyte, called B cells. B cells are good for neutralizing viruses on repeated infection, but a strong response also requires T cells to kill infected cells, to support the B cells and establish immune memory.
Researchers at the Center for Genetic Engineering and Biotechnology in Havana combined DIII with the capsid protein (C) found on all four serotypes, which has effective T-cell epitopes, to produce an aggregate vaccine named DIIIC and asked Fink's group to analyze exactly what sort of immune response it would induce.
Extended trials on immunized mice revealed that the DIIIC vaccine induced a so-called Th1-type response, which helps to promote cytotoxic T cells and regulate the production of antibodies from B cells. Moreover, the antibodies induced by DIIIC were still at useful levels 120 days after immunization.
"In natural dengue infection an imbalance towards a Th2 response appears to be associated with more severe disease and a stronger Th1 response is preferred, so it is encouraging to see an efficient, long-lasting Th1 response to DIIIC," says Fink. Her team hopes to continue collaboration with their Cuban counterparts to understand and develop this promising vaccine for clinical trials using A*STAR's state-of-the-art facilities.
More information: "Tetravalent dengue DIIIC protein together with alum and ODN elicits a Th1 response and neutralizing antibodies in mice." Vaccine 33, 1474–1482 (2015). dx.doi.org/10.1016/j.vaccine.2015.01.074