Overcoming immune exhaustion from chronic HIV infection
Chronic HIV infection results in exhaustion of the immune system, a phenomenon characterized by dysfunctional HIV-specific killer T cells. The exhausted T cells display inhibitory proteins on their surface, and scientists hope to be able to restore immune function by interfering with the negative signals transmitted by such proteins. A study published on December 3rd in PLOS Pathogens suggests that one such strategy blocking the PD-1/PD-L1 pathway works, but only when the overall HIV load is low and regulatory T cells cooperate.
Interaction between the PD-1 protein found on killer T cells and its binding partner PD-1L is one hallmark of immune exhaustion, and interfering with this interaction a potential strategy to overcome it. Based on promising pre-clinical studies, therapeutic trials blocking PD-L1 in HIV-infected patients have started. Besides dampening the response of exhausted killer T cells, however, the PD1/PD-1L pathway also plays a role in the function of regulatory T (or Treg) cells, which themselves suppress the immune response to HIV infection.
Given these complex interactions, Andreas Meyerhans and Jordi Argilaguet, both from the Pompeu Fabra University in Barcelona, Spain, and colleagues argue that it is important to understand the consequences for Treg cells of blocking PD-L1. In this study, the researchers investigated the impact of such blockage on cells from HIV-infected individuals. They isolated blood cells from HIV-positive patients on antiretroviral therapy and at different stages of disease progression, as well as cells from non-infected individuals. They also studied cells taken from HIV-infected individuals before and after they started antiretroviral therapy, and before and after treatment interruptions.
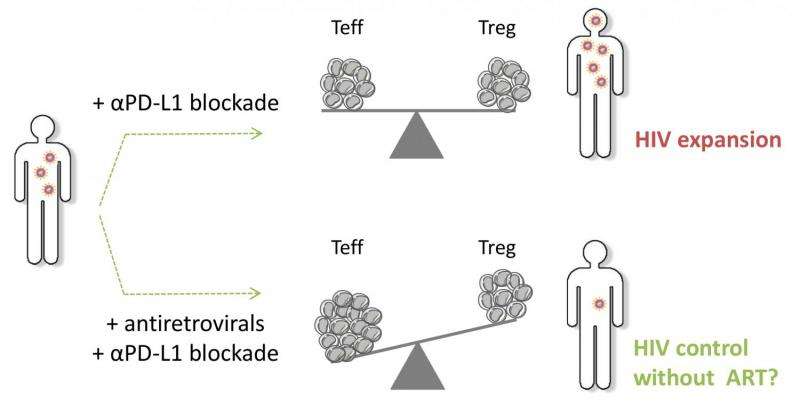
They found that PD-L1 blockade had no effect on the ability of Treg cells to suppress the immune response, regardless of which donor the cells came from. In contrast, PD-L1 blockade increased the capacity of Treg cells to multiply (and hence their overall numbers), but only in cells from patients with viremia, i.e. those that had detectable virus in their blood. In addition, they found that PD-L1 blockade increased virus production in the cell culture from such patients. The higher virus numbers were related to the increase in Treg cell frequencies, suggesting that immune suppression by Treg cells plays a role in virus expansion upon PD-L1 blockade.
Unlike the differential effect on Treg cell proliferation following PD-L1 blockage, the researchers observed an increase in the proliferation of killer T cells from all HIV study groups, regardless of their viral load. Based on these ex vivo experiments (in cells isolated from patients and then exposed to PD-L1 blocking agents outside of the body), they predict that when actual patients are given PD-L1 blocking agents, their viral load at the time will influence the "net" outcome, i.e., whether the blockage boosts or weakens the overall anti-HIV immune response.
While they say their findings support the rationale to combine a PD-L1 blockade with antiretroviral treatment to restore effector responses in HIV infected individuals, the researchers also suggest that the observed differential and plasma viremia dependent gain of Treg cell proliferation and overall suppressive function upon PD-L1 blockade has direct consequences for patient selection to enter clinical trials targeting the PD-1/PD-L1 signaling pathway and treatment modalities.
More information: Peligero C, Argilaguet J, Güerri-Fernandez R, Torres B, Ligero C, Colomer P, et al. (2015) PD-L1 Blockade Differentially Impacts Regulatory T Cells from HIV-Infected Individuals Depending on Plasma Viremia. PLoS Pathog 11(12): e1005270. DOI: 10.1371/journal.ppat.1005270

















